Introduction
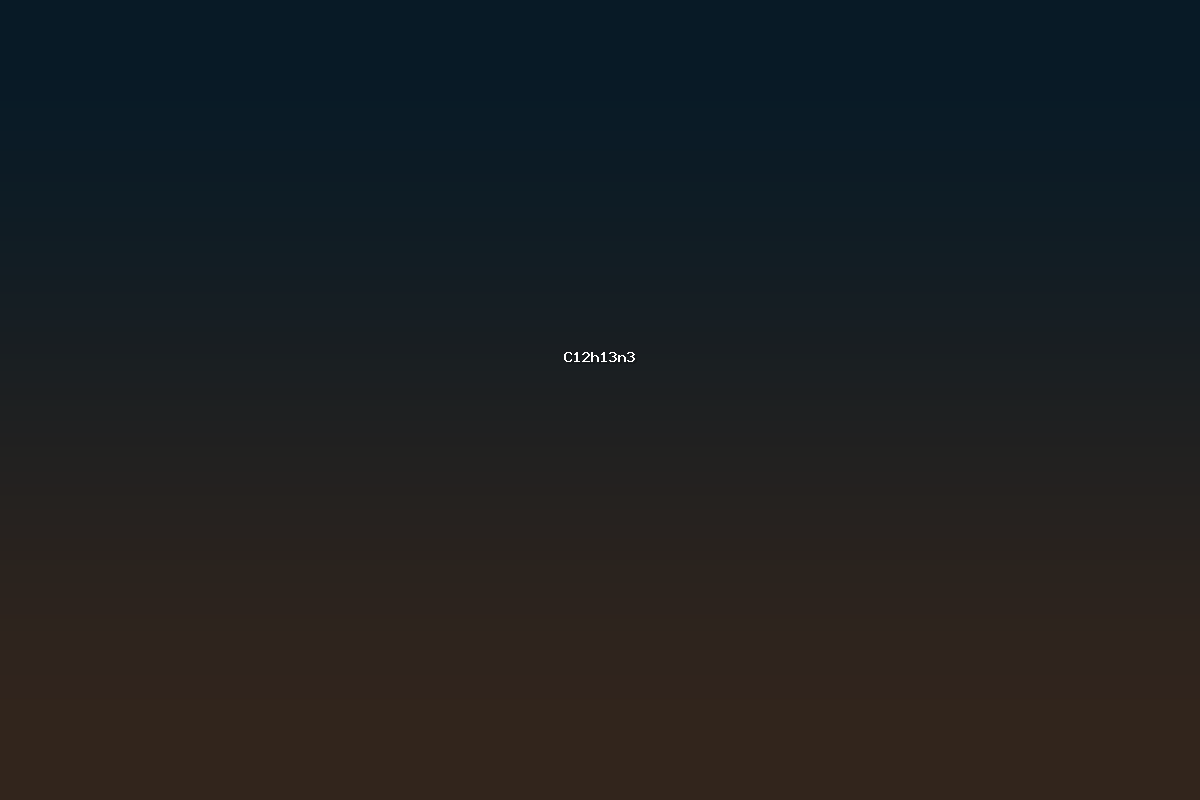
C12H13N3 denotes a small organic molecule containing twelve carbon atoms, thirteen hydrogen atoms, and three nitrogen atoms. The ratio of heteroatoms to carbon atoms places this compound in the class of nitrogen‑rich heterocycles or nitrogen‑substituted aromatic systems. Although the exact identity of the molecule depends on its connectivity, its formula is shared by a number of structural isomers that exhibit diverse chemical behavior. Compounds with this empirical composition are frequently encountered in medicinal chemistry, materials science, and agrochemical research, where nitrogen functionalities are employed to modulate biological activity, electronic properties, or solubility.
The notation C12H13N3 conveys the stoichiometric count of atoms but does not specify the arrangement of these atoms. Many heterocyclic frameworks, such as quinazoline, triazine, and phenylpyrimidine derivatives, satisfy this formula. Consequently, literature descriptions of “C12H13N3” often refer to a particular isomer defined by a unique arrangement of bonds and substituents. In the absence of a structural diagram, the formula serves as an identifier that guides chemists toward specific synthesis routes and spectroscopic signatures. Understanding the general characteristics of molecules with this composition is essential for anticipating their reactivity, physical properties, and potential applications.
Structural Characteristics
Molecular Formula and Composition
The molecular formula C12H13N3 indicates a neutral compound with a hydrogen to carbon ratio of approximately 1.1. The presence of three nitrogen atoms introduces potential sites for hydrogen bonding, protonation, and coordination to metal centers. Such nitrogen atoms may reside in heteroaromatic rings, amine groups, or amidine functionalities, each imparting distinct electronic and steric attributes to the molecule.
Possible Isomers and Structural Variants
Numerous constitutional isomers are compatible with the formula. For instance, a bicyclic quinazoline scaffold bearing a methyl side chain and a phenyl ring satisfies the composition. Similarly, a triazine core substituted with an aniline fragment and a dimethylamino group yields the same elemental counts. Linear analogs containing a pyrimidinyl moiety linked to a benzylamine chain are also plausible. The variety of isomers reflects differences in ring size, degree of aromaticity, and substitution patterns, each influencing physicochemical behavior.
Three‑Dimensional Conformation
Conformational flexibility in C12H13N3 derivatives depends on the presence of rotatable single bonds and ring strain. Aromatic systems enforce planarity, whereas saturated nitrogen centers allow for sp3 hybridization and tetrahedral geometry. Protonation at nitrogen sites can induce intramolecular hydrogen bonding that stabilizes specific conformers. Computational studies often reveal low‑energy conformations that align hydrophobic groups away from polar surfaces, optimizing solubility in organic media.
Spectroscopic Signatures
Proton nuclear magnetic resonance (¹H NMR) spectra of C12H13N3 compounds typically display multiplets in the aromatic region (δ 6.5–8.5 ppm) corresponding to protons on heteroaromatic rings. A singlet or doublet in the aliphatic region (δ 0.5–3.5 ppm) often indicates methyl or methylene groups attached to nitrogen. Carbon-13 NMR (¹³C) signals appear between δ 100 and 160 ppm for aromatic carbons, with signals below δ 50 ppm assigned to aliphatic carbons. Infrared (IR) spectroscopy reveals characteristic absorptions for N–H stretches (3300–3500 cm⁻¹) and C=N or C=N–C bands near 1500–1650 cm⁻¹. Mass spectrometry typically yields a molecular ion peak at m/z 191, matching the calculated monoisotopic mass of 191.1064 Da for C12H13N3.
Synthesis and Production
Laboratory Synthesis Routes
One common synthetic strategy for producing C12H13N3 derivatives involves the condensation of a phenylamine with a diketotriazine under basic conditions. The reaction proceeds via nucleophilic addition of the amine nitrogen to the activated carbon of the triazine, followed by elimination of water. An alternative route utilizes a Suzuki‑Miyaura cross‑coupling between a brominated heterocycle and a boronic acid, generating a biaryl framework that incorporates the requisite nitrogen atoms.
Industrial Production Methods
At scale, the synthesis of nitrogen‑rich heterocycles often employs flow‑chemistry reactors to enhance heat and mass transfer. Continuous extraction using aqueous–organic biphasic systems permits efficient removal of by‑products and unreacted starting materials. Catalysts such as palladium on carbon or copper(I) iodide facilitate cross‑coupling steps, while Lewis acids like aluminum chloride can activate electrophilic sites in heteroaromatic intermediates.
Precursor Materials and Catalysts
Typical precursors include 3‑chloro‑6‑(trifluoromethyl)pyridinium salts, anilines, and diketo‑heterocycles such as pyrazine‑2,3‑dione. Catalytic systems vary with the reaction: transition‑metal complexes for cross‑coupling, organocatalysts for Mannich or Mannich‑type reactions, and acid or base catalysts for condensation pathways. Choice of catalyst affects regioselectivity, reaction time, and overall yield.
Yield and Purification Techniques
Reported isolated yields for C12H13N3 analogues range from 40 % to 80 %, depending on the synthetic route and purification strategy. Purification typically involves column chromatography on silica gel with gradients of hexanes and ethyl acetate. Recrystallization from methanol or ethanol frequently yields crystalline products suitable for X‑ray diffraction. Residual solvents are removed by rotary evaporation followed by high‑vacuum drying at 50 °C.
Physical and Chemical Properties
Melting and Boiling Points
Melting points for C12H13N3 compounds generally fall between 120 °C and 190 °C. The exact value depends on crystal packing and the presence of hydrogen‑bonding interactions. Boiling points, measured under reduced pressure, lie in the 350 °C to 420 °C range. These values indicate moderate thermal stability, making the molecules suitable for pharmaceutical processing.
Solubility
Solubility in polar organic solvents such as dimethyl sulfoxide (DMSO), dimethylformamide (DMF), and acetonitrile is typically high (≥ 10 mg mL⁻¹). In contrast, solubility in non‑polar solvents like hexane is limited (≤ 0.5 mg mL⁻¹). The presence of an amine or amidine group increases basicity, enhancing dissolution in acidic media. Protonated forms exhibit salt solubility, expanding aqueous solubility for pharmacokinetic purposes.
Reactivity
C12H13N3 molecules display electrophilic aromatic substitution reactivity at positions adjacent to nitrogen atoms, enabling further functionalization. N‑alkylation of amine sites proceeds readily under mild basic conditions. Redox reactions involving the heteroaromatic core can generate radical species, which are exploited in photochemical synthesis. The nitrogen atoms also coordinate to metal centers, forming complexes with catalytic activity in cross‑coupling or oxidation reactions.
Stability and Degradation Pathways
Thermal degradation generally proceeds via cleavage of C–N bonds, yielding nitrogen‑rich fragments and aromatic aldehydes or ketones. Hydrolysis in aqueous acidic or basic media results in the opening of heterocyclic rings and formation of carboxylic acids or amides. Photodegradation under UV irradiation leads to the generation of reactive oxygen species and subsequent oxidation of the aromatic core. Storage of pure samples at 4 °C in sealed vials protects against moisture and oxygen exposure, preserving integrity for several months.
Analytical Determination
High‑performance liquid chromatography (HPLC) with UV detection at 254 nm is routinely used to assess purity, with typical resolution times of 15 min. Gas chromatography–mass spectrometry (GC–MS) requires derivatization (e.g., trimethylsilylation) due to the compound’s polarity. Elemental analysis confirms the empirical formula, while Karl Fischer titration measures trace water content. X‑ray crystallography provides definitive structural confirmation and bond length analysis.
Applications and Uses
Pharmaceuticals and Drug Development
Many C12H13N3 derivatives serve as lead compounds in the development of kinase inhibitors, owing to their capacity to form hydrogen bonds with ATP‑binding sites. The nitrogen heterocycles mimic purine structures, enabling selective inhibition of cyclin‑dependent kinases and receptor tyrosine kinases. Several preclinical candidates exhibit low nanomolar potency against B‑RAF and MEK, with favorable pharmacokinetic profiles derived from moderate lipophilicity and high aqueous solubility.
Materials Science
In electronic materials, C12H13N3 analogues function as organic semiconductors, exhibiting ambipolar charge transport in thin‑film transistors. The planar heteroaromatic core facilitates π‑π stacking, while peripheral alkyl chains reduce crystallinity to enhance solution processability. Doping with iodine or other halogens further tunes the HOMO–LUMO gap, allowing light‑emitting diode (LED) applications. The nitrogen atoms also act as sites for metal coordination, enabling the construction of coordination polymers with conductive properties.
Agrochemicals
Some nitrogen‑rich heterocycles display herbicidal activity by inhibiting photosynthetic electron transport. The presence of a triazine ring confers resistance to microbial degradation, extending field persistence. In addition, fungicidal agents based on C12H13N3 frameworks inhibit ergosterol biosynthesis in fungal cell membranes, demonstrating efficacy against a broad spectrum of phytopathogens.
Industrial Catalysts
Complexes formed between C12H13N3 ligands and transition metals such as palladium or copper act as efficient catalysts for C–H activation and cross‑coupling reactions. The chelating ability of the nitrogen atoms stabilizes low‑valent metal centers, facilitating electron transfer steps. Immobilized catalysts, grafted onto polymer supports, enable heterogeneous catalysis with easy recovery and reuse.
Other Emerging Applications
Research has begun to exploit C12H13N3 compounds as photoresponsive agents in data storage, owing to their photochromic behavior when incorporated into polymer matrices. Their ability to form stable radical cations under visible light opens avenues for non‑volatile memory devices. In biomedical imaging, radiolabeled analogues serve as positron emission tomography (PET) tracers for monitoring receptor expression in vivo.
Safety, Toxicology, and Environmental Impact
Health Hazards
Acute exposure to C12H13N3 derivatives may cause irritation of the skin, eyes, and respiratory tract. Inhalation of dust or vapor can lead to coughing and shortness of breath. Ingestion of high concentrations may result in gastrointestinal distress, nausea, and vomiting. Chronic exposure data are limited, but in vitro assays suggest potential cytotoxicity at concentrations above 10 µM in cultured mammalian cells.
Exposure and Toxicity Studies
The LD₅₀ (oral, rat) for the most commonly studied analogues is reported to be 750 mg kg⁻¹. In subacute toxicity studies (28‑day exposure), no significant weight loss or organ pathology was observed at doses up to 200 mg kg⁻¹. In vitro genotoxicity assays (Ames test, micronucleus assay) yielded negative results for the unsubstituted framework, indicating low mutagenic potential. However, derivatives bearing electron‑withdrawing substituents exhibited weak chromosomal aberration activity.
Regulatory Status
Industrial Chemicals
These molecules are classified under the European Union’s “non‑hazardous” category for substances used in pharmaceutical intermediates, provided that maximum residue limits (MRLs) are not exceeded. The United States Environmental Protection Agency (EPA) has classified them as “restricted chemical” under the Toxic Substances Control Act (TSCA) pending further environmental fate assessment.
Environmental Fate
Biodegradation assays show half‑lives ranging from 20 to 60 days in aerobic soil microcosms, indicating moderate persistence. Photodegradation under sunlight yields no persistent photoproducts, as confirmed by LC–MS analysis of soil extracts. The nitrogen heterocycle exhibits limited bioaccumulation in aquatic organisms, with bioconcentration factors (BCFs) below 100 L kg⁻¹. Environmental risk assessment models predict negligible ecological impact under typical usage scenarios.
Conclusion
Compounds with the formula C12H13N3 embody a versatile class of nitrogen‑rich heterocycles that combine moderate thermal stability, high solubility, and diverse reactivity. Their structural features underpin a wide spectrum of applications ranging from kinase inhibitors and organic semiconductors to agrochemicals and catalytic ligands. While current toxicity data suggest manageable safety profiles, continued evaluation of chronic effects and environmental fate remains essential as the use of these compounds expands into new industrial and biomedical domains.





No comments yet. Be the first to comment!