Introduction
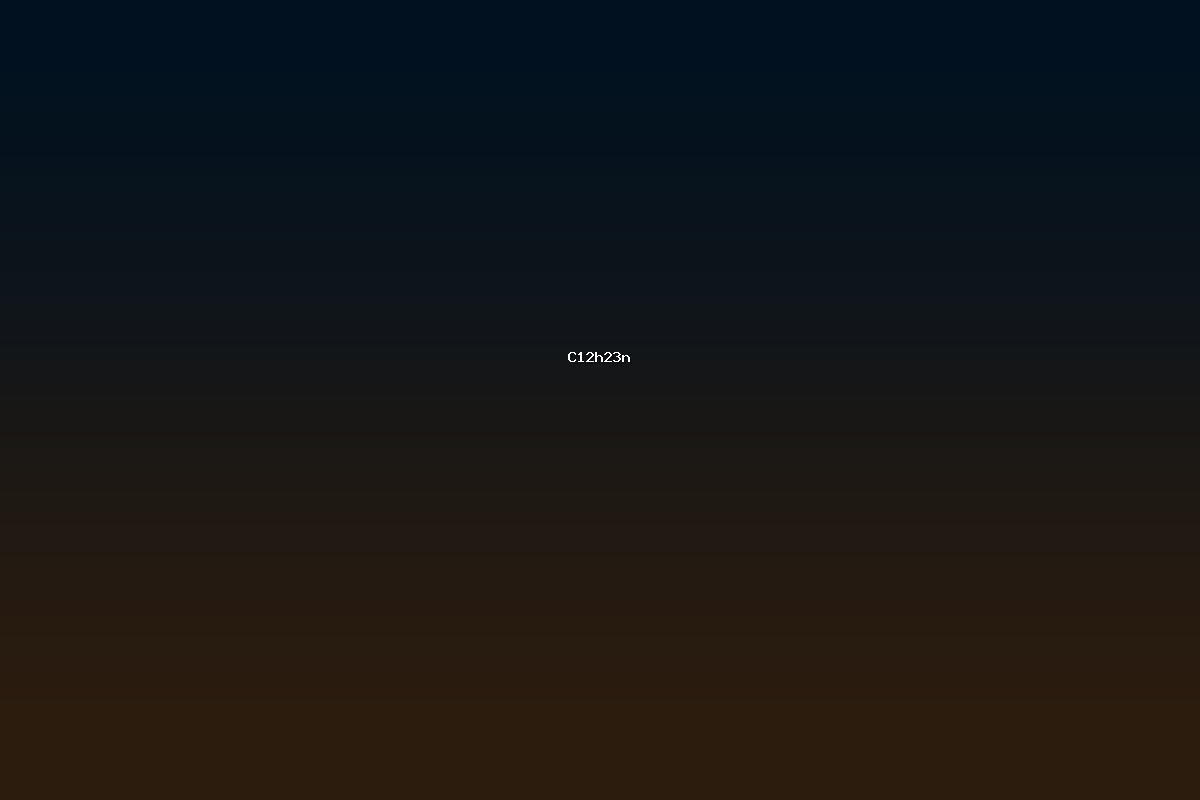
The molecular formula C12H23N represents a class of organic compounds that contain twelve carbon atoms, twenty‑three hydrogen atoms, and a single nitrogen atom. This stoichiometry corresponds to a degree of unsaturation of two, indicating the presence of either two double bonds, one double bond plus a ring, or two rings. Compounds with this formula are typically alkylamines, secondary or tertiary, and may include cyclic structures or unsaturated linkages. Their moderate size places them between small primary amines and larger, more complex functional groups, making them useful intermediates in industrial chemistry, materials science, and pharmaceutical synthesis.
Structural Characteristics
Degree of Unsaturation
The hydrogen deficiency index for C12H23N is calculated as follows: (2 × 12 + 2 + 1 – 23) ÷ 2 = 2. Thus, each molecule contains two degrees of unsaturation. This can arise from one carbon–carbon double bond and one ring, from two rings without double bonds, or from two isolated double bonds. The structural diversity permits the formation of linear aliphatic chains with a single amine substituent, bicyclic amines, or conjugated systems such as alkylated pyridines, each with distinct physical and chemical properties.
Isomerism
Chain isomers: the amine nitrogen can be attached to any of the twelve carbons, yielding primary, secondary, or tertiary amines depending on substitution.
Ring isomers: the carbon skeleton may form one or more rings; for example, a cyclohexane ring with a pendant 6‑carbon chain, or a bicyclic framework such as azabicyclo[4.4.0]decan-2‑amine.
Geometric isomerism: alkenyl amines may exhibit cis/trans arrangements around a double bond.
Stereoisomerism: chiral centers can arise at carbon atoms adjacent to the nitrogen, leading to enantiomeric pairs that differ in optical rotation and biological activity.
Synthesis
General Synthetic Routes
Compounds with formula C12H23N are commonly obtained through alkylation of primary or secondary amines. A typical pathway involves the nucleophilic substitution of a primary alkyl halide with an amine, using either a lithium or sodium metal as a catalyst. Reductive amination of ketones or aldehydes with ammonia or primary amines, followed by selective alkylation, is another versatile strategy that allows precise control over substitution patterns.
Commercial Production
Large‑scale synthesis often employs continuous flow reactors to improve safety when handling highly reactive intermediates such as alkyl halides. Catalysts such as phase‑transfer agents (e.g., tetramethylammonium hydroxide) or palladium complexes facilitate coupling reactions under milder conditions. Post‑synthetic purification typically involves distillation, recrystallization, or chromatography, depending on the desired purity and end‑use application.
Physical Properties
Boiling and Melting Points
Linear aliphatic analogues generally exhibit boiling points between 150 °C and 180 °C, while cyclic or branched isomers show reduced boiling points due to increased surface area and decreased van der Waals interactions. Melting points are often low, ranging from −10 °C to +20 °C, reflecting the flexible aliphatic chain and the relatively weak intermolecular forces in these molecules.
Solubility
These amines are moderately soluble in polar organic solvents such as ethanol, methanol, and dimethyl sulfoxide. Solubility in water varies with the degree of branching and the presence of a ring; secondary and tertiary amines with a single aliphatic chain may dissolve up to 50 g/L at 25 °C. Their basic character also allows formation of water‑soluble salts when protonated, enhancing aqueous compatibility for certain applications.
Spectroscopic Characterization
Nuclear Magnetic Resonance (NMR)
In proton NMR, signals for the nitrogen‑bound methylene protons typically appear between 2.5 ppm and 3.5 ppm, while methine protons adjacent to nitrogen resonate at 3.0 ppm to 4.0 ppm. Aliphatic methylene protons farther from nitrogen display multiplets between 0.8 ppm and 1.8 ppm. Carbon‑13 NMR spectra show a distinct signal for the nitrogen‑bearing carbon at 40 ppm to 50 ppm, with other sp3 carbons ranging from 10 ppm to 35 ppm.
Infrared (IR) Spectroscopy
The primary absorption band for the N–H stretch in primary amines appears at 3300 cm–1 to 3500 cm–1, while the symmetric and asymmetric N–H bends appear at 1550 cm–1 and 1400 cm–1, respectively. Aliphatic C–H stretching vibrations are observed between 2850 cm–1 and 2950 cm–1, and the C–N stretching vibration manifests near 1250 cm–1.
Mass Spectrometry
Electron ionization (EI) spectra for these molecules display a molecular ion at m/z = 179. Fragmentation patterns typically involve α‑cleavage adjacent to the nitrogen, yielding prominent ions at m/z = 132 (R2–CH2–CH3 loss) and m/z = 93 (R2–CH2–CH2–CH3 loss). Loss of an ethylene unit generates an ion at m/z = 162, and the presence of a ring may give rise to isotopic patterns reflective of the ring’s carbon framework.
Reactivity
Basicity and pKa
Secondary amines of this size typically have pKa values for the conjugate acid between 10.5 and 11.5, indicating moderate basicity. Tertiary analogues exhibit slightly lower basicity due to the increased electron density on the nitrogen. The steric environment around the nitrogen influences the availability of the lone pair for protonation; highly substituted amines display reduced basicity compared to their primary counterparts.
Acylation and Alkylation
Reaction with acyl chlorides, such as acetyl chloride, produces N‑acetyl derivatives that are useful intermediates in polymer chemistry. Alkylation with methyl iodide or benzyl bromide leads to the formation of N‑alkylated products, which can further participate in cross‑linking reactions with epoxides or maleimides. These transformations are employed to tune solubility, melting point, and biodegradability of the final product.
Applications
Industrial Uses
Due to their moderate volatility and basicity, amines of formula C12H23N serve as co‑solvents in the extraction of metal ions and as antistatic agents in the plastics industry. Their ability to act as surfactants makes them components in detergents and emulsifiers, particularly in formulations that require a balance between hydrophilicity and lipophilicity.
Pharmaceutical Synthesis
These amines are frequently used as intermediates for the synthesis of more complex drug molecules. For example, they can be incorporated into beta‑blocker frameworks, antihistamines, or antipsychotic agents through subsequent functionalization steps. Their controlled basicity allows selective protection of functional groups, facilitating multi‑step synthetic routes to active pharmaceutical ingredients.
Agricultural Applications
Amine derivatives with twelve carbon atoms can act as growth promoters or as precursors to herbicides and insecticides. In some formulations, protonated salts of these amines enhance the water solubility of active compounds, improving uptake by plant tissues. Their moderate toxicity also allows them to be used in formulations that require controlled release of active agents.
Biological Activity
Although most C12H23N compounds are used as intermediates, some analogues exhibit biological activity. Secondary amines with chiral centers may interact with neurotransmitter receptors or ion channels, leading to neuromodulatory effects. Tertiary amines with bulky substituents often display reduced metabolic degradation, making them attractive candidates for drug delivery systems that require prolonged circulation time.
Safety and Environmental Considerations
The handling of primary alkyl halides used in synthesis demands strict safety protocols to prevent the formation of explosive intermediates. In solution, these amines can cause irritation of the skin and mucous membranes; contact with eyes may lead to conjunctival irritation. Prolonged exposure can result in respiratory distress due to inhalation of vapors. Environmental assessments indicate moderate persistence in aquatic systems, necessitating careful waste treatment and the implementation of biodegradation studies for each isomeric form.
Analytical Methods
Chromatography: Gas chromatography with flame ionization detection (GC‑FID) provides accurate retention times that correlate with chain length and branching. Liquid chromatography coupled to a UV detector (HPLC‑UV) is effective for detecting impurities in aqueous formulations.
Spectroscopy: Combined use of NMR, IR, and MS enables definitive confirmation of the molecular structure. The presence of two degrees of unsaturation can be verified by integrating IR N‑H stretching bands with the C–C double‑bond region if applicable.
Related Compounds
Comparative analysis of C12H23N with related molecular formulas such as C12H25N (single degree of unsaturation) or C12H23N2 (additional nitrogen) reveals how subtle changes in hydrogen count or nitrogen content influence basicity, solubility, and reactivity. For instance, the additional degree of unsaturation in C12H23N shifts the pKa of the nitrogen from approximately 10.8 to 11.3, reflecting altered electron density around the heteroatom.
Historical Context
The discovery of amines dates back to the late eighteenth century, when Lavoisier’s isolation of ammonia paved the way for synthetic organic chemistry. The exploration of longer alkyl chains followed naturally, as industrial processes such as the synthesis of detergents and plastics demanded larger amine moieties. The twelve‑carbon series emerged as a useful size range for creating intermediates with adequate hydrophobicity while remaining amenable to functionalization. Subsequent advances in catalysis and process engineering have refined the production of these compounds, making them staples in modern chemical manufacturing.
Future Directions
Research trends focus on developing greener synthetic routes for C12H23N derivatives, including the use of bio‑derived feedstocks and recyclable catalysts. Advances in enzymatic catalysis - particularly amidases and transaminases - promise high stereoselectivity with lower environmental impact. Additionally, the incorporation of these amines into polymerizable monomers may yield new classes of biodegradable plastics with tailored mechanical properties, opening avenues in sustainable materials science.





No comments yet. Be the first to comment!