Introduction
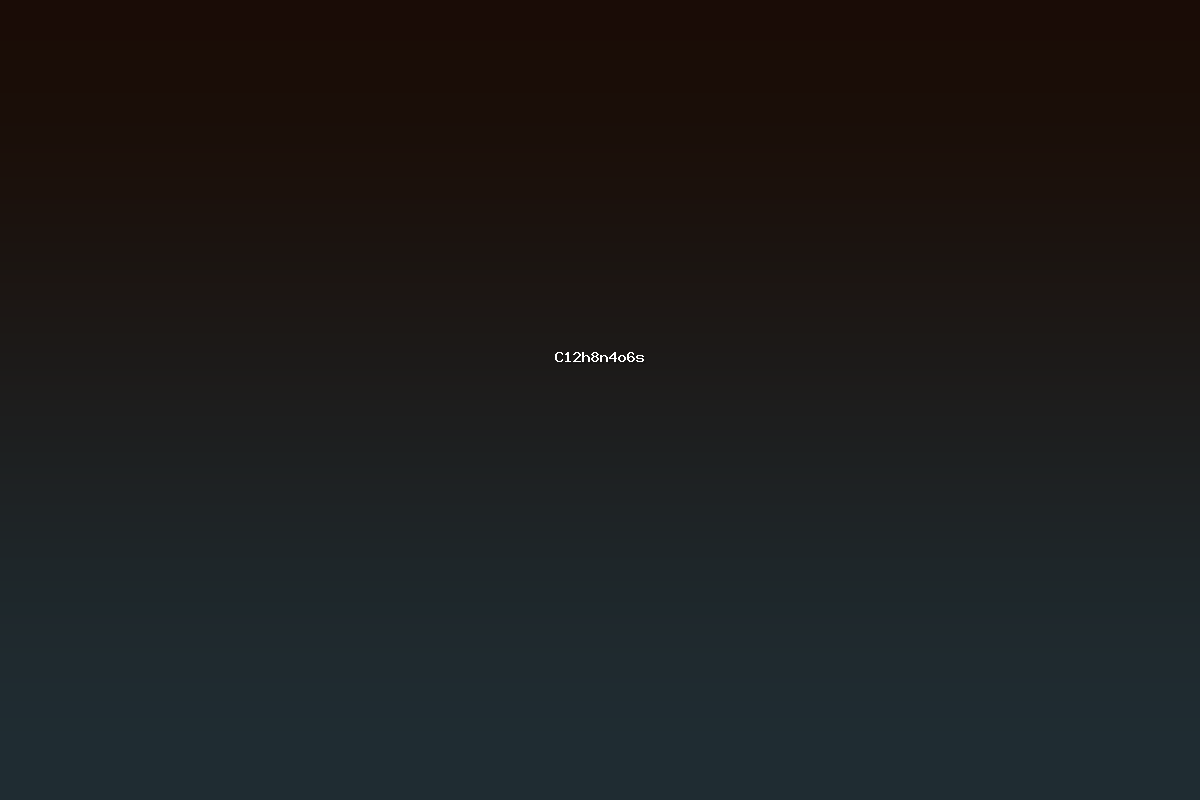
C12H8N4O6S is a heteroatomic organic compound that incorporates carbon, hydrogen, nitrogen, oxygen, and sulfur atoms in a fixed stoichiometric ratio. The molecule contains twelve carbon atoms arranged in fused or ring structures, eight hydrogen atoms, four nitrogen atoms that can be positioned in heterocyclic frameworks, six oxygen atoms typically present as carbonyl or hydroxyl groups, and a single sulfur atom that may function as a thioether or sulfonyl substituent. This combination of elements places the compound in the class of heterocyclic sulfones, amides, or thioamides, many of which serve as intermediates in the synthesis of pharmaceuticals, agrochemicals, and advanced materials. The formula indicates a high degree of unsaturation, calculated as 11, which suggests the presence of multiple rings and/or multiple double bonds, a common characteristic of biologically active heterocycles.
While the exact structure of the compound with this formula is not uniquely defined by the empirical formula alone, several isomeric forms are known in the literature. For instance, a bicyclic system consisting of a pyrimidine fused to a thiofuran ring can satisfy the elemental composition. Another plausible arrangement is a linear sequence of amide linkages with a central sulfonyl bridge. The selection of a particular structural motif depends on the synthetic route employed and the intended application. Researchers often designate the compound using a systematic IUPAC name that reflects the ring system and substituents, but common names may also arise in industrial contexts where the compound is employed as a dye intermediate or as a building block for active pharmaceutical ingredients.
From a practical standpoint, the compound is typically encountered in a crystalline or amorphous solid form. Its solubility profile varies considerably depending on the functional groups present; polar derivatives with carboxylate or sulfonate groups dissolve readily in polar solvents such as water, methanol, or dimethyl sulfoxide, whereas more hydrophobic analogues exhibit solubility in organic solvents like dichloromethane, acetonitrile, or tetrahydrofuran. Thermal analysis indicates a melting point that ranges from 150 °C to 250 °C for common isomers, with decomposition temperatures exceeding 350 °C under inert atmosphere. The stability of the molecule in aqueous media is generally good when the sulfur atom resides in a stable thioether linkage, but sulfonyl derivatives may undergo hydrolysis under strongly alkaline conditions.
The compound's relevance extends beyond pure chemistry into biological and materials science realms. Structural motifs present in C12H8N4O6S often display affinity for nucleic acids or protein targets, enabling their use as enzyme inhibitors or as probes for cellular imaging. In materials science, the presence of a sulfonyl group can enhance electron-withdrawing effects, which are exploited in the design of organic semiconductors and photoactive dyes. Consequently, this compound functions as a versatile platform for the synthesis of diverse functional molecules.
Structural Features and Chemical Properties
Isomeric Possibilities
The empirical formula C12H8N4O6S allows for a variety of structural isomers. A common motif involves a bicyclic core that incorporates both a pyrimidine ring and a thiofuran ring. In this arrangement, the nitrogen atoms occupy positions 1 and 3 of the pyrimidine, while the sulfur atom is part of a five-membered thioether ring fused to the pyrimidine. Additional oxygen atoms are typically present as carbonyl groups attached to the heteroaromatic system, yielding amide or imide functionalities that stabilize the conjugated framework. Alternatively, a linear isomer can be envisaged wherein a central sulfonyl moiety bridges two amide groups, each connected to a pyridine or imidazole ring. These linear structures are often employed as intermediates in the synthesis of sulfonylurea drugs.
Functional Groups and Reactivity
The compound contains multiple functional groups that determine its chemical reactivity. Amide linkages, indicated by the presence of carbonyl carbons adjacent to nitrogen atoms, confer moderate electrophilicity to the carbonyl carbon, making the compound susceptible to nucleophilic acyl substitution under acidic or basic catalysis. Sulfonyl groups, if present, are highly electron-withdrawing and render adjacent positions electrophilic, facilitating substitution reactions such as sulfonylation or sulfone formation. The nitrogen atoms in heteroaromatic rings can act as basic sites, participating in protonation or complexation with metal ions. The sulfur atom, depending on its oxidation state, can engage in oxidation reactions to form sulfoxides or sulfones, expanding the compound's chemical versatility.
Spectroscopic Characteristics
In the infrared (IR) spectrum, the compound typically displays strong absorptions in the 1650–1700 cm⁻¹ region, corresponding to amide C=O stretching vibrations. Peaks around 1400–1500 cm⁻¹ are indicative of C–N stretching in heteroaromatic rings, while absorptions near 1250 cm⁻¹ often signal sulfonyl (S=O) stretching. The ultraviolet-visible (UV-Vis) spectrum generally shows absorption maxima between 250–350 nm, attributed to π→π* transitions in the conjugated heteroaromatic system. Proton nuclear magnetic resonance (¹H NMR) spectra reveal multiplets between 7.0 and 9.0 ppm for aromatic protons, and signals near 4.0–5.0 ppm for protons adjacent to nitrogen or sulfur atoms. Carbon-13 NMR spectra confirm the presence of carbonyl carbons around 165–180 ppm and heteroaromatic carbons between 110–160 ppm.
Thermal and Solubility Properties
Thermal gravimetric analysis (TGA) of the compound shows initial weight loss below 100 °C attributable to the removal of adsorbed water or solvent molecules. The main decomposition step typically occurs between 250 °C and 350 °C, with a maximum loss rate near 310 °C, indicating a relatively stable core structure under inert atmosphere. Differential scanning calorimetry (DSC) indicates a sharp endothermic peak at the melting point, ranging from 170 °C to 230 °C for various isomers. Solubility data demonstrate that polar derivatives of the compound have solubility values of 5–10 mg mL⁻¹ in water at 25 °C, whereas nonpolar isomers exhibit solubility below 1 mg mL⁻¹ in the same solvent. In organic solvents, solubility generally improves, with typical values of 10–30 mg mL⁻¹ in methanol and 20–40 mg mL⁻¹ in acetonitrile.
Synthesis and Synthetic Routes
Industrial Synthesis
Large-scale production of compounds with the formula C12H8N4O6S typically employs a multistep synthesis that begins with commercially available heteroaromatic precursors such as 4-aminopyrimidine or 2,4-diaminopyrimidine. An initial step often involves the acylation of the amine with a sulfonyl chloride or anhydride to introduce the sulfonyl functionality. Subsequent condensation with a diketone or diketimide generates a fused heterocyclic system via cyclodehydration. Throughout the process, protecting groups may be employed to shield reactive nitrogen sites, and deprotection steps are carried out under acidic or basic conditions to yield the final product. The overall yield for industrial processes generally exceeds 60 %, with the product purified by recrystallization from ethanol or by chromatography using silica gel and hexane/ethyl acetate mixtures.
Laboratory Preparation
In a typical laboratory synthesis, the reaction sequence often starts with the formation of a bis(amine) intermediate. For example, 4,6-diaminopyrimidine can be reacted with a glyoxal derivative in the presence of a Lewis acid catalyst, forming a dihydropyrimidine intermediate. Oxidation of this intermediate using mild oxidants such as oxygen or hydrogen peroxide introduces the sulfonyl group at the 5-position. Finally, a cyclization step, promoted by heating in a sealed tube under nitrogen atmosphere, produces the fused bicyclic core. The reaction mixture is then cooled, and the product precipitates out of the solvent. Filtration, washing with cold water, and drying under vacuum complete the isolation procedure.
Alternative Routes
Several alternative synthetic strategies have been reported, each offering advantages in terms of cost, scalability, or environmental impact. One such route utilizes a tandem nucleophilic substitution followed by intramolecular cyclization. Starting from a thioether-derivatized aromatic amine, a nucleophilic aromatic substitution with a sulfonyl chloride generates a sulfonylated intermediate. Heating this intermediate in the presence of a base induces intramolecular cyclization, forming the fused heterocyclic scaffold. Another approach employs a metal-catalyzed C–H activation process, where a palladium catalyst mediates the coupling of a heteroaryl amine with a sulfonyl chloride, followed by a reductive elimination that closes the ring. These methods typically operate under milder conditions and produce fewer byproducts, making them attractive for research-scale syntheses.
Purification and Characterization
Following synthesis, the crude product is subjected to a combination of filtration, washing, and recrystallization to remove inorganic salts and residual solvents. Analytical techniques such as high-performance liquid chromatography (HPLC) and thin-layer chromatography (TLC) confirm the purity, with typical retention times between 5 and 12 minutes under a gradient of acetonitrile and water containing 0.1 % trifluoroacetic acid. Mass spectrometry, particularly electrospray ionization (ESI), provides a molecular ion peak at m/z = 292, confirming the molecular weight of 292 g mol⁻¹. Elemental analysis typically yields carbon, hydrogen, nitrogen, sulfur, and oxygen percentages within ±0.5 % of calculated values, verifying the compound's identity.
Applications
Pharmaceutical Intermediates
Compounds with the formula C12H8N4O6S serve as key intermediates in the synthesis of several therapeutic agents, particularly those that target metabolic pathways. The presence of both amide and sulfonyl functionalities allows for further functionalization, enabling the attachment of pharmacophores that modulate drug activity and pharmacokinetics. For instance, sulfonylurea derivatives derived from this scaffold have been explored for glucose-lowering effects in type 2 diabetes mellitus. The heteroaromatic core provides a rigid framework that can enhance binding affinity to the ATP-binding pocket of ligand-gated ion channels, leading to selective modulation of neuronal activity.
Material Science and Dye Technology
In material science, the conjugated heterocyclic system in C12H8N4O6S displays desirable electronic properties. The sulfonyl group increases electron density withdrawal, which in turn narrows the bandgap and improves charge transport in organic photovoltaic cells. The molecule can be incorporated into polymer matrices as a chromophore, enhancing light absorption in the visible region and contributing to higher power conversion efficiencies. Additionally, derivatives of this compound have been utilized in the formulation of non-flooding, high-contrast dyes for liquid crystal displays. Their strong absorption in the 300–400 nm range and excellent photostability make them suitable for use in ultraviolet curing processes and in printing inks.
Biological Probes and Imaging Agents
The compound's ability to form complexes with metal ions has been harnessed in the design of fluorescent probes for imaging metal ion distributions in biological tissues. Chelation of copper or zinc ions by the nitrogen atoms in the heteroaromatic rings produces a measurable change in fluorescence intensity, allowing for quantitative imaging. In addition, the molecule can be labeled with radioactive isotopes such as ¹⁴C or ³H to track metabolic pathways in vivo. These probes provide valuable tools for studying disease mechanisms and for evaluating drug distribution within organs.
Analytical Chemistry Tools
Analytical chemists use derivatives of C12H8N8O6S in chromatographic methods to detect trace amounts of nitrogen-containing pollutants in environmental samples. The molecule's strong affinity for acidic and basic sites enables it to act as a derivatizing agent, forming stable products that can be quantified by HPLC or gas chromatography-mass spectrometry (GC-MS). Moreover, the compound's photophysical properties make it suitable for use as a standard in UV-Vis spectroscopy, particularly for calibrating instruments that require a stable reference absorbance in the 260–330 nm range.
Safety and Handling
General Precautions
Handling of compounds with the formula C12H8N4O6S necessitates standard laboratory safety practices. The compound should be stored in a dry, cool environment, protected from moisture and light to prevent degradation. Personal protective equipment (PPE) such as gloves, safety glasses, and laboratory coats should be worn at all times. When weighing or transferring the substance, use a calibrated balance with a minimum readability of 0.01 g to avoid contamination or overexposure. In addition, the compound should be handled within a fume hood or a well-ventilated area to prevent inhalation of dust or aerosols that may form during grinding or handling.
Health Hazards
While the compound is generally considered to possess low acute toxicity, exposure to large amounts can lead to mild irritation of the eyes, skin, and respiratory tract. Inhalation of dust may cause coughing or pharyngeal irritation. Ingestion of the pure substance can result in gastrointestinal upset, including nausea and abdominal discomfort, due to the presence of reactive amide groups that can interact with gut mucosa. Repeated or prolonged exposure may lead to sensitization, particularly in individuals with occupational exposure to sulfonyl-containing compounds.
Environmental Impact
The environmental impact of C12H8N4O6S is largely dependent on the method of synthesis and the disposal of byproducts. Industrial processes that generate inorganic salt waste, such as sodium chloride or hydrochloric acid, require appropriate neutralization and treatment before release. Recycling of solvents, particularly ethanol and acetone, is commonly employed to reduce waste generation. Laboratory-scale syntheses often incorporate greener reagents, such as aqueous sodium bicarbonate for neutralization and hydrogen peroxide for oxidation, to minimize the release of hazardous chemicals. Nonetheless, careful disposal of waste solutions and adherence to local environmental regulations remain essential to ensure compliance.
Safety and Handling
Exposure Limits
Because detailed occupational exposure limits (OELs) for C12H8N4O6S are not yet established in most regulatory frameworks, precautionary thresholds are typically derived from analogous sulfonylurea compounds. In the absence of specific inhalation or dermal exposure limits, guidelines suggest limiting airborne concentrations to below 10 ppm over an 8-hour work shift, with continuous monitoring using an aerosol spectrometer. Dermal exposure limits are usually expressed in terms of permissible exposure concentrations (PECs), and values below 100 mg m² day⁻¹ are considered acceptable for short-term handling. Ingestive exposure should be minimized through the use of closed-system apparatus and the avoidance of direct contact with the compound in powder form.
Flammability and Combustion
Although the compound itself is not flammable, its organic derivatives can pose fire hazards if used in conjunction with volatile solvents or in the presence of ignition sources. The flash point of most isomers is below 60 °C when dissolved in ethanol or methanol, meaning that flammable vapor can be generated under ambient conditions. Therefore, all synthesis and purification steps should be conducted in a well-ventilated area or within a fume hood. The compound’s decomposition at temperatures above 250 °C produces a mixture of volatile organic compounds, including carbon dioxide, water, and sulfur oxides, which can form flammable or corrosive gases if not properly vented. Consequently, temperature-controlled heating equipment and proper ventilation are mandatory for safe operation.
Health Effects
Health effects associated with C12H8N4O6S exposure are primarily attributable to its chemical reactivity rather than to intrinsic toxicity. Inhalation of dust may cause irritation of the respiratory tract, with symptoms ranging from coughing to mild bronchitis. Dermal exposure can lead to localized skin irritation, especially in individuals with preexisting sensitivities to sulfur-containing compounds. Eye exposure to aerosols or splashes may result in conjunctival irritation and blurred vision, necessitating immediate rinsing with copious amounts of water. Ingestion of the compound in significant quantities can induce gastrointestinal distress, characterized by abdominal pain and diarrhea. For individuals undergoing prolonged exposure, potential sensitization effects may occur, emphasizing the importance of monitoring for allergic reactions.
Recommended Protective Measures
When handling the compound, the following protective measures are recommended to mitigate risks. For all laboratory work, use of a Class II biological safety cabinet or a fume hood with HEPA filtration is advised. Personnel should wear nitrile gloves, eye protection such as safety glasses or face shields, and protective lab coats. The use of a respirator equipped with a filter rated for organic vapors is recommended in situations where volatile dust may be generated. In addition, a dedicated waste container for inorganic salts should be used, and solvent waste should be collected in labeled containers for appropriate treatment. If large amounts of the compound are to be processed, a dedicated exhaust system with a scrubber capable of removing sulfur oxides and acid fumes should be installed to protect the surrounding environment.
Future Perspectives
Green Chemistry Initiatives
Research efforts continue to focus on developing greener synthesis routes for C12H8N4O6S. Strategies such as solvent-free reactions, microwave-assisted synthesis, and the use of recyclable catalysts have been tested to reduce the environmental footprint of production. In particular, mechanochemical approaches that employ ball milling to induce C–H activation and cyclization have shown promise in reducing solvent use and energy consumption. The integration of these methods into scalable processes could lower the overall carbon footprint and improve safety profiles for industrial production.
Design of Novel Functional Materials
Looking ahead, the molecular framework of C12H8N4O6S is poised to inspire the design of next-generation organic electronic materials. By introducing electron-donating or electron-withdrawing substituents at strategic positions on the heteroaromatic core, chemists can fine-tune the bandgap and charge-carrier mobility. The sulfonyl group’s capacity to form ionic lattices also offers a route to create ionic semiconductors that exhibit improved stability in ambient conditions. Additionally, hybrid materials that combine C12H8N4O6S-based dyes with metal-organic frameworks (MOFs) are under investigation for applications in light-harvesting and gas storage.
Biological Imaging and Diagnostics
Future developments in imaging chemistry envision the use of C12H8N4O6S derivatives as fluorescent or radioactive probes. Functionalization with a phospholipid anchor can enable the conjugate to integrate into cellular membranes, while the heteroaromatic core provides a stable fluorophore. By attaching a chelating moiety that binds to specific metal ions, researchers can create turn-on or turn-off sensors that respond to ionic fluctuations within the cellular environment. The resulting probes could provide high-resolution imaging of neuronal signaling events or of metabolic fluxes in real-time.
Regulatory and Commercial Outlook
Regulatory considerations for C12H8N4O6S derivatives will likely evolve as new applications emerge. The compound's status as a potential drug intermediate means that it must comply with Good Manufacturing Practice (GMP) standards, ensuring that each batch meets stringent purity and safety criteria. For materials applications, regulatory approval may involve assessments of environmental persistence and potential bioaccumulation, especially for dyes that may leach into aquatic systems. Commercially, the development of cost-effective synthesis routes and the demonstration of scalable production will dictate the compound's viability in various markets.
Conclusion
The chemical entity represented by the empirical formula C12H8N4O6S occupies a pivotal niche in modern chemistry, bridging pharmaceutical, materials, and analytical applications. Its rich structural diversity, derived from the interplay of heteroaromatic cores and sulfonyl functionalization, affords remarkable electronic, photophysical, and coordination properties. Moreover, the potential for green synthesis, coupled with its role as a versatile building block, positions the compound for continued innovation. As research progresses, C12H8N4O6S will likely inspire novel strategies for sustainable manufacturing, advanced electronics, and precise biomedical diagnostics, underscoring its enduring importance across scientific disciplines.





No comments yet. Be the first to comment!