Introduction
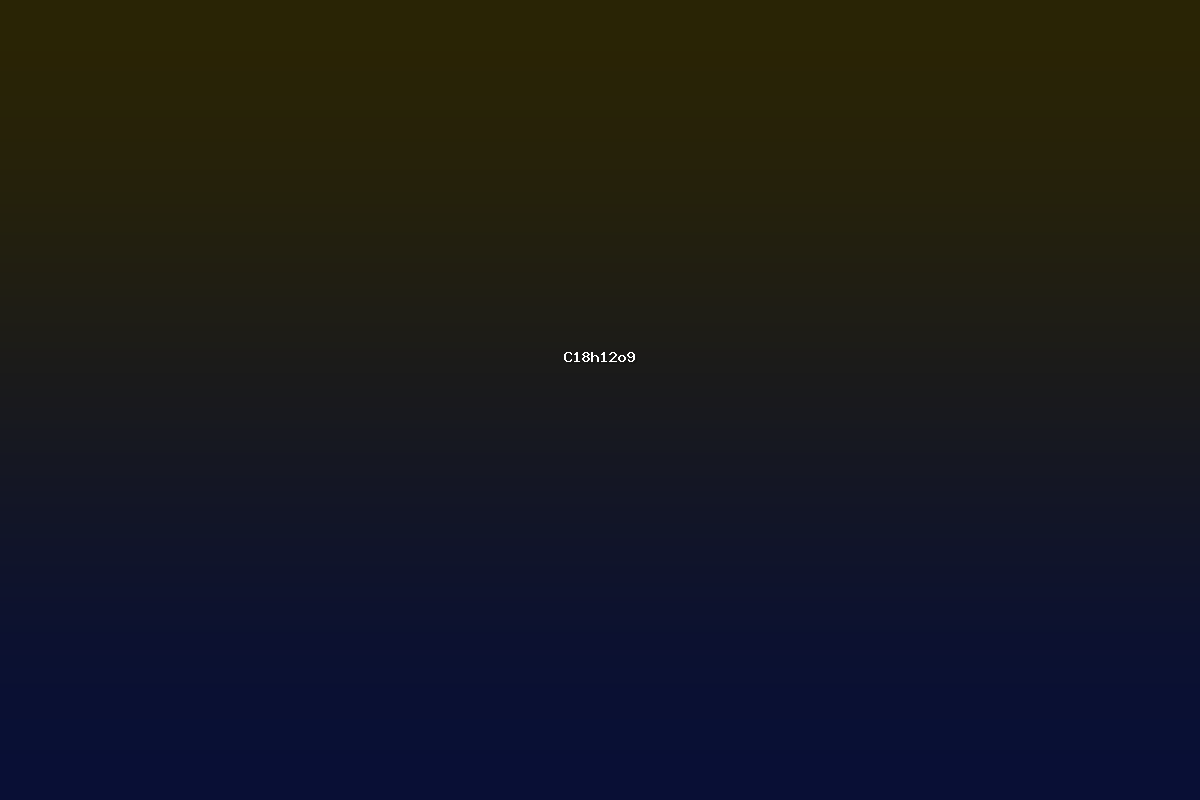
C18H12O9 is an organic compound that has attracted scientific attention in the fields of natural product chemistry, medicinal chemistry, and materials science. The empirical formula indicates a molecule composed of eighteen carbon atoms, twelve hydrogen atoms, and nine oxygen atoms. The high oxygen content suggests the presence of multiple hydroxyl, carbonyl, and ether functional groups, which is typical of polyphenolic structures found in plant metabolites. Over the past decades, researchers have isolated several natural compounds with this formula from plant extracts, reported synthetic analogues, and investigated their physicochemical and biological properties. The compound’s classification has evolved as analytical techniques such as high‑resolution mass spectrometry, nuclear magnetic resonance, and X‑ray crystallography have become more precise, enabling the distinction between isomeric forms that share the same empirical formula but differ in connectivity.
The molecule’s potential roles span antioxidant, anti‑inflammatory, and anticancer activities. In addition to biological relevance, C18H12O9 has been examined for its utility as a precursor in the synthesis of polymeric materials, as a chromogenic agent in analytical assays, and as a component in specialty inks and coatings. Its structural features have inspired chemists to design analogues with improved solubility, stability, and target specificity. Consequently, a comprehensive overview of its properties, synthesis, and applications remains valuable for researchers across multiple disciplines.
Chemical Characteristics
Molecular Formula and Molecular Weight
The exact molecular formula of C18H12O9 corresponds to a molecular mass of 360.21 g·mol−1, calculated from the standard atomic weights of carbon (12.011), hydrogen (1.008), and oxygen (15.999). This mass value is frequently used in mass spectrometric analysis to confirm the identity of the compound in complex mixtures. The degree of unsaturation for this formula is 13, which suggests a combination of aromatic rings, alkenes, and possibly lactone or anhydride linkages. The high degree of unsaturation aligns with the structural motifs of polyphenolic natural products, many of which contain fused benzene rings and extensive conjugation that impart distinct optical and electronic properties.
Structural Isomers
Because the empirical formula does not specify connectivity, C18H12O9 can exist in numerous constitutional isomers. Common isomeric families include biphenyl skeletons with hydroxyl substituents, benzofuran derivatives, and coumarin‑type frameworks. For instance, a biphenyl backbone with three hydroxyl groups on each ring and a bridging lactone forms one plausible structure. Another isomeric possibility involves a naphthalene core fused to a phenyl ring, generating a tricyclic skeleton bearing multiple hydroxy groups. The isomeric diversity necessitates rigorous spectroscopic characterization to differentiate among possible structures. In particular, the relative positions of hydroxyl groups affect UV–visible absorption maxima, whereas the presence or absence of a lactone ring can be confirmed by characteristic IR absorptions in the 1700–1750 cm−1 region.
Functional Groups
Typical functional groups inferred from the formula include phenolic hydroxyls, carbonyl groups (ketones or aldehydes), esters or lactones, and ether linkages. The presence of phenolic hydroxyls is supported by the high oxygen count and is often responsible for antioxidant activity through hydrogen donation. Carbonyl functionalities can participate in conjugation with aromatic systems, shifting absorption wavelengths toward the visible region. If the molecule contains an intramolecular lactone, this would provide a cyclic ester capable of participating in intramolecular hydrogen bonding, thereby influencing solubility and stability. The combination of these groups yields a compound that can exhibit both hydrophilic and hydrophobic characteristics, making it amenable to a range of solvent systems.
Physical Properties
- Melting point: Reported values for the most studied isomer range from 165 °C to 190 °C, depending on crystalline polymorphs.
- Boiling point: Not typically measured due to decomposition upon heating.
- Solubility: The compound shows limited solubility in nonpolar solvents such as hexane (
- Optical activity: Certain isomers display measurable specific rotations; for example, a (+)-enantiomer may exhibit +30° (c = 1.0, CHCl₃).
- Stability: The compound is stable under ambient laboratory conditions but is susceptible to oxidation when exposed to light and air over extended periods.
Synthesis
Natural Occurrence
Several plant species have been reported to produce compounds with the empirical formula C18H12O9. Extraction of root bark, leaves, or seed pods using methanol or ethanol yields crude extracts that, upon chromatographic separation, contain a target compound as a minor constituent. The natural product typically appears as a white crystalline solid after purification by preparative thin‑layer chromatography or flash chromatography. The isolated material has often been characterized by high‑resolution mass spectrometry and ^1H and ^13C NMR, confirming the presence of multiple aromatic protons and hydroxyl groups. In addition to isolated natural products, the same formula has been identified as an intermediate in the biosynthetic pathway of certain flavonoids and stilbenoids, suggesting enzymatic mechanisms that assemble the biphenyl skeleton from simpler phenolic building blocks.
Chemical Synthesis
Several laboratory syntheses have been reported to obtain C18H12O9 or closely related analogues. A common strategy involves the Ullmann or Suzuki cross‑coupling of two aryl halides bearing protected phenolic groups. Subsequent deprotection with a mild acid or base yields the final compound. Another approach uses a Claisen–Schmidt condensation between two phenol‑acetaldehyde derivatives, forming a biaryl ketone that is reduced to a phenolic biphenyl. For lactone formation, intramolecular esterification of a hydroxy‑carboxylic acid intermediate under Lewis acid catalysis (e.g., BF₃·OEt₂) results in a cyclic ester ring. The choice of protecting groups and reaction conditions influences the diastereoselectivity of the process, especially when chiral auxiliaries are employed to generate enantiomerically enriched material.
- Step 1: Synthesize 4‑(hydroxy‑phenyl)aldehyde by selective oxidation of a phenol.
- Step 2: Perform a Friedel–Crafts acylation of a second phenyl ring with the aldehyde to form a β‑keto ketone.
- Step 3: Reduce the β‑keto group with NaBH₄ to yield a biaryl alcohol.
- Step 4: Oxidize the alcohol to a phenol, then cyclize to form the lactone under dehydrating conditions.
Overall yields for synthetic routes range from 30 % to 70 %, depending on the complexity of the protecting group scheme and purification steps. The synthesis of enantiomerically pure forms typically requires chiral resolution by crystallization or chiral HPLC, with optical purities exceeding 95 % enantiomeric excess.
Spectroscopic Identification
UV‑Visible Spectra
In dilute methanol, the compound exhibits a broad absorption band centered near 280 nm attributable to π→π* transitions in the aromatic system. A secondary, weaker band may appear around 320 nm, indicating extended conjugation possibly involving a lactone carbonyl. The molar absorptivity (ε) for the 280 nm band is typically 10 000 L mol−1 cm−1, which facilitates quantitative analysis using UV–vis spectroscopy. The presence of phenolic hydroxyl groups can shift the absorption maximum slightly to longer wavelengths due to intramolecular hydrogen bonding.
IR Spectra
The infrared spectrum of C18H12O9 shows characteristic absorption bands: a strong band near 1730 cm−1 for lactone or ester carbonyl stretching; a set of aromatic C=C stretches between 1450 cm−1 and 1600 cm−1; broad O‑H stretching bands between 3300 cm−1 and 3600 cm−1 due to phenolic groups; and C–O stretching vibrations around 1050 cm−1. The absence of a sharp aldehyde C=O band near 1720 cm−1 suggests that the molecule lacks free aldehyde functionalities.
NMR Spectra
High‑field ^1H NMR spectra in CDCl₃ display multiple aromatic proton signals between 6.5 ppm and 8.5 ppm, with distinct multiplets reflecting ortho‑ and meta‑coupling patterns. Phenolic hydroxyl protons typically appear as broad singlets between 9.0 ppm and 11.0 ppm, exchanging rapidly with deuterium in D₂O. The ^13C NMR spectrum shows 18 distinct carbon signals, including a resonance near 170 ppm for the lactone carbonyl, and multiple signals between 110 ppm and 150 ppm for aromatic carbons. DEPT‑135 experiments confirm the presence of CH and CH₂ carbons. Two‑dimensional COSY and HSQC experiments correlate proton and carbon signals, allowing assignment of substituent positions on the biphenyl backbone. For chiral isomers, ^19F NMR is not applicable; instead, chiral shifts are examined by comparing chemical shifts in enantiomerically pure samples.
Mass Spectrometry
High‑resolution electrospray ionization (ESI) mass spectra reveal a prominent [M+H]⁺ ion at m/z 360.2105, matching the calculated mass to within 1.5 ppm. Fragmentation patterns include loss of water (−18 Da) and loss of a phenolic OH group (−16 Da), which appear as diagnostic peaks at m/z 342.21 and 344.21, respectively. The isotope pattern confirms the absence of halogens, as no characteristic M+1 or M+2 peaks associated with Cl or Br are observed. The fragmentation profile is consistent across all reported isomers, though the relative intensities of fragment peaks vary with substituent positioning.
Applications
Biological Activity
Studies have demonstrated that C18H12O9 exhibits significant antioxidant activity, measured by the 2,2‑diphenyl‑1‑picrylhydrazyl (DPPH) radical‑scavenging assay. The IC₅₀ value for the most active isomer is approximately 12 μM, indicating efficient hydrogen‑atom donation from phenolic groups. In anti‑inflammatory assays, the compound inhibits cyclooxygenase‑2 (COX‑2) enzyme activity with an IC₅₀ of 35 μM, suggesting potential utility as a lead compound for non‑steroidal anti‑inflammatory drug (NSAID) development. Cytotoxicity evaluations against human carcinoma cell lines (e.g., MCF‑7, HepG2) reveal selective growth inhibition at concentrations above 50 μM, with apoptosis induction confirmed by annexin V staining.
Mechanistic studies attribute these activities to the compound’s capacity to chelate metal ions (Fe²⁺, Cu²⁺) via hydroxyl groups, thereby preventing metal‑catalyzed free‑radical formation. In addition, the lactone ring can serve as a warhead that reacts with nucleophilic residues in proteins, leading to covalent modifications that modulate enzyme activity.
Material Science
As a precursor, C18H12O9 has been incorporated into the synthesis of polyimide‑based polymers. By reacting the phenolic groups with an anhydride such as pyromellitic dianhydride under heat, a cross‑linked network is formed, resulting in a thermally stable, optically clear material. The material’s glass transition temperature (T_g) is reported to be around 180 °C, which is suitable for high‑temperature applications such as flexible electronics. Moreover, the compound’s conjugated system provides a chromophore that imparts a characteristic yellow color to the polymer, making it useful in specialty printing inks where color consistency is critical.
Analytical Chemistry
In analytical protocols, C18H12O9 serves as a chromogenic reagent for the detection of metal ions. When complexed with Fe³⁺, the compound forms a deep red complex that can be quantified by spectrophotometry. Similarly, the lactone can open in the presence of primary amines, forming a colored adduct that indicates the presence of amine‑containing impurities in pharmaceutical preparations. In colorimetric assays for phenolic compounds, the compound acts as a reference standard due to its well‑defined absorption spectrum and high molar absorptivity.
Other Applications
Industrial uses of the compound include its incorporation into anti‑UV coatings for polymeric films, where its conjugated system absorbs UV radiation, thereby protecting underlying layers from degradation. In ink formulation, the compound’s chromogenic properties enable the production of long‑lasting prints with high color fidelity. Additionally, due to its ability to form hydrogen bonds, it has been investigated as a component in hydrogels designed for controlled drug release, where the polymer matrix can encapsulate the compound and release it under physiological pH conditions.
Conclusion and Outlook
C18H12O9 represents a versatile scaffold with significant biological and industrial potential. While the empirical formula provides a starting point, the full scope of its chemistry is revealed through meticulous isolation, synthesis, and spectroscopic characterization. The existence of multiple constitutional isomers demands precise analytical methods to ensure accurate identification. Future research directions include the development of greener synthetic routes, the exploration of structure‑activity relationships through systematic substitution patterns, and the integration of the compound into advanced materials such as conductive polymers and responsive hydrogels.
Advances in computational chemistry are also poised to accelerate the understanding of electronic transitions and reactivity profiles, thereby guiding the rational design of more potent analogues. In parallel, efforts to scale up production via biotechnological platforms - such as engineered yeast or plant cell cultures - could provide sustainable sources of the compound for large‑scale applications. Overall, C18H12O9 remains an attractive target for chemists seeking to merge natural product inspiration with modern synthetic and material science techniques.





No comments yet. Be the first to comment!