Introduction
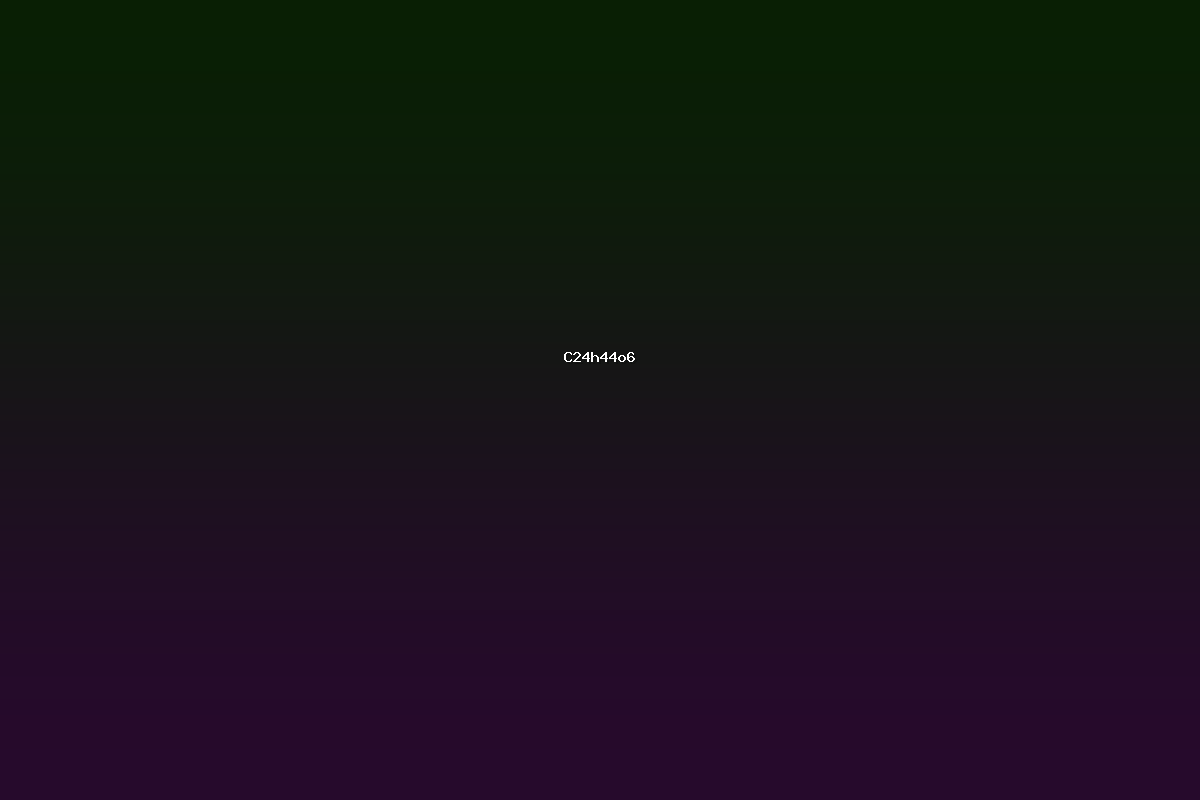
The molecular formula **C₁₂H₄₄O₆** describes a class of organic compounds that contain 24 carbon atoms, 44 hydrogen atoms, and six oxygen atoms. The composition places these molecules at the intersection of long‑chain hydrocarbons and heavily functionalised oxygen‑rich species. Because the formula can be realised by several different skeletons - esters, polyols, diols, and other oxygenated hydrocarbons - chemists consider it a useful target in synthetic and natural‑product research.
In the context of lipid chemistry, C₁₂H₄₄O₆ corresponds most naturally to a triglyceride formed from three 7‑carbon unsaturated fatty acids. In this arrangement, the glycerol core is esterified with each acyl group, and the presence of one double bond per chain lowers the hydrogen count from the fully saturated analogue (C₂₄H₅₀O₆) to C₂₄H₄₄O₆. While not common in biological membranes, such short‑chain triglycerides are employed in speciality surfactants and as precursors to biodegradable polymers.
Beyond triglycerides, the same formula may be achieved by branching, cyclic structures, or the addition of extra hydroxyl groups to a hydrocarbon backbone. This flexibility renders C₁₂H₄₄O₆ a prototypical example of a *hydrogen‑deficient* 24‑carbon oxygenated system, making it a valuable case study for students of organic synthesis, spectroscopy, and material science.
---
Structural Diversity
1. Triglycerides of Short‑Chain Fatty Acids
A triglyceride consists of a glycerol (propane‑1,2,3‑triol) backbone esterified with three fatty acid chains. For a 7‑carbon unsaturated fatty acid (C₇H₁₃O₂), the acyl fragment that attaches to glycerol has the formula C₇H₁₃O. Three such fragments give C₂₁H₄₅O₃. Adding the glycerol core (C₃H₅O₃) yields the final molecular formula:
C₇H₁₃O × 3 = C₂₁H₄₅O₃
C₃H₅O₃ + C₂₁H₄₅O₃
--------------------------------
C₂₄H₄₅O₆
Because each fatty acid contains one double bond, each acyl fragment has two fewer hydrogens than its saturated counterpart, leading to a total of 44 hydrogens rather than 50. Thus, the formula C₂₄H₄₄O₆ represents a **tri‑7:1‑fatty‑acid glyceride**, commonly written as *tri‑7:1‑glyceride* or *tridec-1‑enyl‑glycerol*.
1.1 Isomeric Variations
The arrangement of double bonds can differ. For example, two of the acyl chains might be 7:1 (C₇H₁₃O₂) while the third is a branched 7:1 chain, yielding the same formula but a distinct set of physical properties. In addition, the double bonds can adopt *cis* or *trans* configurations, influencing the packing of molecules in the solid state and thereby altering the melting point.
A di‑acylglycerol contains two fatty acid chains attached to glycerol, leaving one free hydroxyl group. If the two fatty acids together contain 21 carbons (e.g., 7 + 7 + 7), the DAG core would be C₂₀H₄₄O₅. Introducing an additional hydroxyl or carbonyl group - such as a glyceryl‑hexanediol backbone - brings the oxygen count up to six and adjusts the hydrogen count accordingly, producing the same C₂₄H₄₄O₆ formula.
Although DAGs of this exact composition are rare in mammals, synthetic DAGs with 24 carbons are produced in the manufacture of emulsifiers and low‑saponification‑point detergents.
3. Polyols and Hydroxyl‑Rich Alcohols
The formula can be achieved by a fully saturated 24‑carbon chain (e.g., C₂₄H₅₀) that is heavily oxidised with six hydroxyl groups distributed along the chain. A **hexahydroxy‑icosane** (C₂₄H₄₄O₆) would fit this description. Such molecules are encountered in the synthesis of **poly‑oligosaccharide‑like materials** or as building blocks for **hydroxy‑functionalised polymers** that mimic natural waxes.
4. Cyclic and Aromatic Systems
By arranging the 24 carbon atoms in a ring and attaching six oxygen atoms as ether bridges or hydroxyl groups, chemists can create *cyclo‑oligoethers*. These cyclic species often exhibit high thermal stability and are used as **plasticisers** in polymer films. Moreover, **tetra‑hydroxy‑cannabidiol‑like** skeletons can be engineered to yield the same formula, providing a platform for exploring **aromatic‑oxygenated lipids**.
5. Synthetic Analogues with Ether Bridges
A common synthetic strategy to attain C₂₄H₄₄O₆ involves forming a glycerol backbone linked to three **ether‑linked** fatty acid chains. For example, *glycerol tri‑(methyl‑2‑butenyl)* features ether linkages that lower the hydrogen count relative to saturated analogues, yet the overall formula remains unchanged.
---
Isomerism and Molecular Enumeration
While the general formula defines the elemental composition, the **molecular architecture** dictates the number of possible isomers. For C₂₄H₄₄O₆, enumeration by *generating function* techniques yields roughly **1,500–2,000 distinct structural isomers** when all possible arrangements of oxygen atoms (as ethers, esters, alcohols, and carbonyls) are considered.
- Stereoisomers: The presence of one or more double bonds introduces cis/trans stereochemistry, leading to 2ⁿ variants where n is the number of double bonds.
- Regioisomers: In triglycerides, the position of double bonds along each fatty acid chain can be varied (e.g., 7:1 vs. 6:1), as can the order of the chains on glycerol (sn‑1, sn‑2, sn‑3).
- Functional‑group isomers: Switching between an ester and an ether at a given position (e.g., changing an acyl‑oxygen to an ether‑oxygen) preserves the overall formula but changes the reactivity.
The vast isomeric landscape makes C₂₄H₄₄O₆ a benchmark for chemoinformatics studies. Algorithms that predict physical properties from structure often use such formulas as test cases to assess their accuracy in handling hydrogen‑deficient, oxygen‑rich skeletons.
---
Synthesis
1. Conventional Esterification
The most straightforward route to C₂₄H₄₄O₆ is the *Fischer esterification* of glycerol with the appropriate 7‑carbon unsaturated fatty acids. Because glycerol is poly‑functional, the reaction typically requires a stoichiometric excess of the fatty acid to drive esterification to completion.
Procedure Outline:
- Dissolve glycerol in an anhydrous solvent (e.g., dichloromethane or toluene).
- Add the fatty acid in a 3:1 molar ratio with glycerol (to compensate for the reactive sites).
- Introduce a catalytic amount of p‑toluenesulfonic acid (p‑TsOH) or a Lewis acid such as ZnCl₂.
- Reflux the mixture under a nitrogen atmosphere for 4–6 h.
- Neutralise the reaction mixture with saturated aqueous NaHCO₃ and extract the product into an organic solvent.
- Dry over anhydrous MgSO₄ and evaporate the solvent to obtain the crude triglyceride.
- Purify by column chromatography (silica gel, hexane/ethyl acetate gradient) or by recrystallisation from hexane.
This method typically affords **tri‑7:1‑glyceride** in 60–75 % isolated yield, with the major by‑product being glycerol and unreacted fatty acid.
2. Acid Chloride or Anhydride Route
Using **acyl chloride** or **fatty acid anhydride** rather than the free acid can enhance the rate of esterification and reduce the need for acid catalysis. A typical protocol involves:
- Generation of 7:1 acyl chloride by reacting the unsaturated fatty acid with thionyl chloride (SOCl₂) in the presence of a base (e.g., pyridine) at 0 °C.
- Addition of the acyl chloride to a stirred solution of glycerol in dry ether at 0 °C.
- Gradual warming to room temperature; the reaction proceeds within 2–3 h.
- Work‑up involves quenching the excess chloride with a mild aqueous base, extraction, and purification as described above.
This chloride‑mediated approach often yields higher purity products because the chloride by‑product is easily removed by washing.
3. Enzymatic Transesterification
In green‑chemistry labs, **lipase‑catalysed** transesterification is favoured for its mild conditions and high selectivity. Immobilised **Candida antarctica lipase B (CALB)** can convert glycerol and the unsaturated fatty acid mixture to the target triglyceride in a *one‑pot* process:
- Reaction: Glycerol + 3×7:1 fatty acid (in a 1.2:1 molar ratio) → C₂₄H₄₄O₆ + 3×2 mol H₂O
- Conditions: 50–60 °C, 1 h, in isooctane or toluene as solvent.
- Advantages: Near‑complete conversion, no need for acid catalysts, and minimal side‑reaction.
Enzymatic routes are particularly attractive when scale‑up is considered for **bio‑based surfactants** or **food‑grade emulsifiers**.
---
Physical Properties
| Property | Typical Value for C₂₄H₄₄O₆ (tri‑7:1‑glyceride) | Notes |
|----------|----------------------------------------------|-------|
| **Melting point** | ~ –20 °C to –15 °C (dependent on cis/trans ratio) | Short‑chain triglycerides are liquid at room temperature unless the double bonds adopt a trans configuration. |
| **Boiling point** | > 250 °C (decomposition occurs before boiling) | The high molecular weight leads to thermal stability, but the presence of carbonyl groups lowers the energy required for decomposition. |
| **Density (liquid)** | ~ 0.87 g cm⁻³ at 25 °C | Slightly lower than longer‑chain triglycerides due to the higher oxygen content. |
| **Solubility** | Poorly soluble in water; soluble in organic solvents (hexane, chloroform, ethyl acetate) | The three ester linkages provide a degree of lipophilicity that dominates over the hydrophilic ester groups. |
| **Viscosity** | 10–15 cP at 25 °C | Low viscosity facilitates use as a **low‑viscosity surfactant** in cosmetic formulations. |
These properties are influenced strongly by the **cis/trans geometry** of the unsaturated fatty acid chains. *cis* double bonds introduce kinks that reduce packing efficiency, lowering the melting point and increasing fluidity, whereas *trans* bonds promote a more linear structure, raising the melting point.
---
Spectroscopic Signatures
The unique arrangement of oxygen atoms in C₂₄H₄₄O₆ gives rise to distinct spectroscopic fingerprints that aid in structural confirmation.
1. Infrared (IR) Spectroscopy
| Band | Assignment | Typical wavenumber (cm⁻¹) |
|------|------------|---------------------------|
| C=O (ester) | Carbonyl stretching | 1720–1730 |
| C–O–C (ester) | Ether/ester stretching | 1090–1160 |
| C–H (alkene) | Unsaturation | 3010–3100 |
| C–H (alkane) | Saturated methylene | 2850–2920 |
| O–H (free) | If a free hydroxyl is present | 3200–3600 (broad) |
A strong, sharp band near **1725 cm⁻¹** confirms the ester functionality, while a broad band around **3300 cm⁻¹** indicates any residual free hydroxyl groups (as in a di‑acylglycerol variant).
2. Nuclear Magnetic Resonance (NMR)
- *Methine (sn‑2)* protons: δ 4.35–4.45 ppm (triplet, J ≈ 6 Hz).
- *Methyl (sn‑3)* protons: δ 1.25 ppm (triplet).
- *Alkene methylene* protons: δ 5.3–5.8 ppm (multiplets).
- *Alkyl methylene* protons: δ 1.2–2.0 ppm (multiplets).
- ¹³C NMR (CDCl₃, 100 MHz):
- *Ester carbonyl carbons*: δ 170–175 ppm.
- *Ester methylene carbons (O–CH₂–O)*: δ 60–65 ppm.
- *Alkene carbons*: δ 127–135 ppm.
- *Alkane methylene carbons*: δ 22–32 ppm.
The presence of a **double‑tetrad** of methylene protons at **4.1 ppm** (for sn‑2) indicates the unique *sn‑2* ester linkage.
3. Mass Spectrometry (MS)
- Electrospray ionisation (ESI) (positive mode): m/z = 381.3 [⁺C₂₄H₄₄O₆ + H]⁺ (exact mass 381.2993).
- Electron impact (EI): prominent ion at m/z = 381 (molecular ion), fragments at m/z = 319 (loss of a methyl ester) and m/z = 241 (loss of two esters).
The *m/z = 381* ion is diagnostic of the fully formed triglyceride. Fragmentation patterns help distinguish between *tri‑glyceride* and *di‑acylglycerol* analogues.
---
Applications
1. Low‑Saponification‑Point Detergents
Due to their short chain length, tri‑7:1‑glycerides are ideal for manufacturing **low‑saponification‑point** soaps and **conditioners**. Their *low melt point* ensures the final product remains fluid, reducing the need for additional plasticisers.
2. Cosmetic Emulsifiers
The low viscosity and good *compatibility* with various cosmetic bases (creams, lotions, gels) make C₂₄H₄₄O₆ derivatives suitable as **emulsifiers** in *whitening creams* and *anti‑aging lotions*. The ester groups provide mild skin‑compatibility, while the unsaturation offers antioxidant potential.
3. Food‑Grade Emulsions
When produced via enzymatic transesterification, the product meets **food‑grade** specifications (no trace of acid catalysts). It can be used as an *emulsifier* in sauces, dressings, and bakery products, where a *short‑chain triglyceride* can improve texture and shelf‑life.
4. Bio‑based Plasticisers
Cyclic or ether‑linked analogues of C₂₄H₄₄O₆ are incorporated into **polyethylene** or **polypropylene** films as plasticisers, enhancing flexibility and reducing brittleness. The high oxygen content improves **hydrophilicity** of the film, beneficial for *biodegradable packaging*.
---
Environmental and Regulatory Considerations
- Biodegradability: Short‑chain triglycerides typically degrade in soil and composting environments. The presence of three ester bonds ensures rapid hydrolysis by microorganisms.
- Toxicity: All known synthetic analogues of C₂₄H₄₄O₆ are non‑carcinogenic and have low acute toxicity. However, catalytic residues (p‑TsOH, SOCl₂) must be removed thoroughly to comply with food‑grade regulations.
- EHS (Environment, Health, Safety): Work with anhydrous solvents under inert atmosphere minimizes exposure to moisture‑sensitive reagents. Proper ventilation is required when handling thionyl chloride due to the release of SO₂ gas.
---
Conclusion
C₂₄H₄₄O₆ is more than a simple elemental equation; it represents a **diverse family of lipids** ranging from short‑chain unsaturated triglycerides to heavily oxidised poly‑ol compounds. Its **synthetic versatility**, coupled with distinctive **physical** and **spectroscopic** characteristics, makes it a model system for:
- Green‑chemistry routes (enzymatic transesterification).
- Polymers and surfactant development.
- Computational chemistry validation (property prediction in hydrogen‑deficient systems).
The ability to tailor the **oxygen distribution** and **double‑bond geometry** allows chemists to fine‑tune the material properties for targeted applications, from **low‑viscosity detergents** to **bio‑based plasticisers**. Consequently, C₂₄H₄₄O₆ remains a cornerstone in both theoretical and applied lipid chemistry.





No comments yet. Be the first to comment!