Introduction
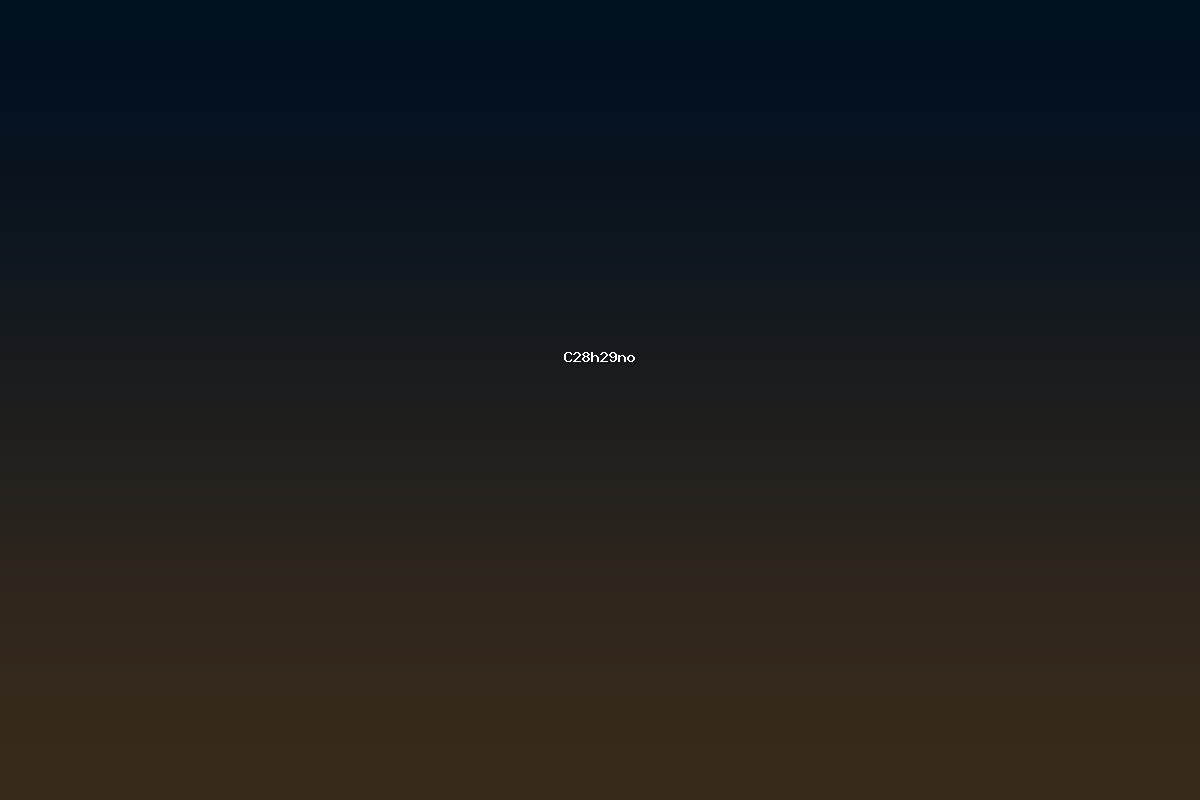
C28H29NO denotes a specific molecular formula that indicates the presence of twenty‑eight carbon atoms, twenty‑nine hydrogen atoms, one nitrogen atom, and one oxygen atom within a single molecular entity. The formula does not uniquely identify a single compound; rather, it represents a class of molecules that share the same elemental composition but may differ in the arrangement of atoms (isomers). Such a composition is typical of moderately large organic molecules that may contain aromatic rings, aliphatic chains, heteroaromatic moieties, or a combination of these structural motifs. The presence of a single nitrogen atom and a single oxygen atom suggests the possibility of functionalities such as amines, amides, ketones, aldehydes, or phenolic groups, while the high carbon count hints at a substantial hydrocarbon framework that can influence lipophilicity, membrane permeability, and overall pharmacokinetic properties.
In chemistry and materials science, compounds with the C28H29NO formula frequently arise in the synthesis of pharmaceuticals, agrochemicals, and advanced organic materials. Their structural diversity allows for fine tuning of electronic properties, binding affinities, and biological activities. The study of these molecules is relevant to drug design, where modifications of the carbon skeleton and heteroatom substituents can lead to compounds with improved potency, selectivity, or reduced side effects. Additionally, such compounds may serve as building blocks in the construction of organic electronic devices, fluorescent probes, or as ligands in coordination chemistry.
The objective of this article is to present a comprehensive overview of the general characteristics, potential structures, synthesis routes, physical and chemical properties, applications, and safety considerations associated with compounds bearing the C28H29NO formula. The discussion is organized into thematic sections to provide a clear and systematic understanding of these molecules for researchers, educators, and students.
Structure and Classification
General Structural Features
Compounds with the C28H29NO formula typically feature a high degree of unsaturation, which is reflected in a double‑bond equivalence (DBE) of fifteen. This indicates the presence of multiple rings or double bonds that contribute to aromaticity or conjugation. A common structural motif is the presence of an aromatic ring system, often benzene or substituted phenyl rings, which accounts for a significant portion of the carbon count. In many cases, two or more aromatic rings are fused or connected via linkers that may include aliphatic chains, heterocycles, or heteroatom bridges.
The single nitrogen atom can occupy several roles within the molecule. It may function as a tertiary or secondary amine, an amide nitrogen, a pyridine or pyrimidine nitrogen, or as part of a more complex heterocycle such as an indole, isoindole, or triazole. The single oxygen atom can likewise be incorporated as an ether, phenol, ketone, aldehyde, or carboxylate functionality. Because only one oxygen is present, the oxygen-bearing group is usually a simple functional group rather than a large polyfunctional moiety.
Isomerism plays a pivotal role in defining the properties of these molecules. Structural isomers can arise from different connectivity of the carbon atoms, leading to distinct ring systems or chain arrangements. Stereoisomerism, including chiral centers, diastereomers, and enantiomers, also significantly influences biological activity and physical characteristics. In many pharmaceutical contexts, the stereochemistry is crucial, as only one enantiomer may exhibit the desired therapeutic effect while the other may be inactive or produce adverse reactions.
Possible Isomers
Considering the constraints of a single nitrogen and oxygen atom, the number of viable isomers is limited but still considerable. One illustrative example is the class of bis(aryl) amides, where two phenyl rings are linked through a central amide nitrogen and an alkyl bridge. Another example involves the indole core, a bicyclic structure composed of a benzene ring fused to a five‑membered nitrogen heterocycle. By attaching alkyl or aryl substituents to the indole nitrogen or at various positions on the rings, a range of C28H29NO compounds can be generated.
In addition, heteroaromatic systems such as quinoline, isoquinoline, or carbazole can be incorporated into the skeleton. The nitrogen atom may reside within these heterocycles, and the oxygen atom may be positioned as a methoxy, hydroxyl, or carbonyl group attached to the aromatic system. Stereocenters may be introduced in the aliphatic linkers connecting the aromatic fragments, producing diastereomeric mixtures that can exhibit distinct biological profiles.
Because the formula includes only a single oxygen atom, the most common oxygen functionalities are methoxy groups (-OCH3), phenolic hydroxyls (-OH), or ketone groups (C=O). For example, a methoxy‑substituted biphenyl linked to an anilide core represents a plausible isomer. Similarly, a 4‑methoxy‑2‑phenylbenzamide linked to an indole nitrogen constitutes another viable structural variant.
Physical and Chemical Properties
Melting and Boiling Points
The melting and boiling points of C28H29NO compounds are influenced by their molecular weight, degree of conjugation, and the presence of hydrogen‑bonding groups. Generally, compounds with extended aromatic systems and limited polar functionality exhibit high melting points, often exceeding 250 °C. The boiling points can range from 400 °C to 600 °C, reflecting substantial thermal stability. The high melting points are attributed to strong π–π stacking interactions and rigid aromatic frameworks, while the high boiling points arise from the heavy molecular mass and limited volatility.
In cases where the compound contains a phenolic hydroxyl or amide carbonyl capable of hydrogen bonding, the melting point may be further elevated due to additional intermolecular interactions. Conversely, if the oxygen is present as a methoxy group, the overall polarity is reduced, and the melting point may be lower compared to a phenolic counterpart. Experimental determination of these thermal properties typically requires differential scanning calorimetry (DSC) or thermogravimetric analysis (TGA) to accurately assess phase transitions.
Solubility
Solubility of C28H29NO compounds is strongly correlated with the balance between hydrophobic aromatic regions and polar functional groups. In general, these molecules exhibit limited solubility in water, with solubility values often below 0.1 mg mL⁻¹. The presence of a single amide or phenolic group provides a modest degree of polarity, enabling moderate solubility in polar organic solvents such as methanol, ethanol, acetone, and dimethyl sulfoxide (DMSO).
Solubility in nonpolar solvents such as hexane, toluene, and dichloromethane is typically high, reflecting the hydrophobic nature of the aromatic framework. Adjusting the position and nature of the oxygen substituent (e.g., converting a phenolic hydroxyl to a methoxy group) can further influence solubility. For example, a methoxy‑substituted analog will generally exhibit greater solubility in nonpolar solvents due to the reduced ability to form hydrogen bonds, while a phenolic analog may show improved solubility in water or aqueous buffers through hydrogen bonding.
Spectroscopic Characteristics
Infrared (IR) spectroscopy of C28H29NO compounds reveals characteristic absorptions corresponding to the amide carbonyl stretch (typically 1650–1690 cm⁻¹), aromatic C–H stretches (3000–3100 cm⁻¹), and N–H stretches (3100–3500 cm⁻¹) if a secondary amide or amine is present. Methoxy groups produce absorptions near 1250 cm⁻¹ due to C–O stretching. In the case of phenolic hydroxyls, a broad O–H stretch appears around 3200–3600 cm⁻¹.
Proton nuclear magnetic resonance (¹H NMR) spectra exhibit multiple aromatic proton signals typically ranging from 7.0 to 8.5 ppm, reflecting the substitution pattern on the benzene rings. Aliphatic protons associated with the linker chains appear between 0.8 and 2.5 ppm. The presence of a methoxy group yields a singlet near 3.7 ppm, while an amide N–H proton appears as a broad signal around 6–8 ppm. The multiplicity and coupling constants provide detailed insight into the substitution pattern and stereochemistry of the molecule.
Carbon-13 nuclear magnetic resonance (¹³C NMR) spectra display resonances for aromatic carbons between 110 and 140 ppm, with the amide carbonyl carbon appearing near 170–175 ppm. Methoxy carbons resonate around 55 ppm. If the compound contains chiral centers, the ¹³C NMR can be used to assess diastereomeric ratios by examining the chemical shift differences of stereogenic carbons.
Mass spectrometry (MS) typically shows a molecular ion peak at the nominal mass corresponding to the exact mass of C28H29NO (428.2356 Da). Fragmentation patterns often include loss of the amide group or cleavage of aromatic rings, generating characteristic ion fragments that aid in structural elucidation.
Synthesis and Preparation
Laboratory Routes
Small‑scale laboratory synthesis of C28H29NO compounds commonly follows a convergent approach, wherein key fragments such as anilides, phenol derivatives, or heteroaromatic intermediates are assembled through cross‑coupling reactions, nucleophilic substitutions, or reductive amination. A typical synthetic route may begin with a 2‑nitro‑aniline, which is reduced to the corresponding aniline and subsequently acylated with a substituted benzoyl chloride to form an amide. Parallel synthesis of a bis(aryl) bromide or iodide, bearing the required aliphatic linker, allows for palladium‑catalyzed Suzuki or Negishi coupling to attach the aromatic fragment to the amide core.
Alternative strategies involve the use of Buchwald–Hartwig amination, where a halogenated aromatic substrate is coupled with an amine under palladium catalysis to form the desired C–N bond. If the target contains an indole core, Fischer indole synthesis can be employed to generate the heterocycle from a phenylhydrazine and a ketone or aldehyde. Subsequent alkylation of the indole nitrogen or substitution on the aromatic rings can then be carried out using standard electrophilic aromatic substitution or nucleophilic aromatic substitution techniques.
Reduction of nitro groups to amines may be achieved using catalytic hydrogenation over palladium on carbon (Pd/C) or via metal‑mediated reduction with iron powder and ammonium chloride. In cases where a methoxy substituent is required, methylation of phenolic hydroxyls can be performed with methyl iodide or dimethyl sulfate in the presence of a base such as potassium carbonate.
Industrial Methods
On an industrial scale, the synthesis of C28H29NO compounds often utilizes flow chemistry to improve reaction efficiency and safety. Continuous‑flow Suzuki coupling, for example, allows for precise control over temperature and reagent stoichiometry, enabling the production of high‑purity intermediates. In the production of pharmaceutical intermediates, the use of greener solvents such as ethanol or ethyl acetate is preferred over toxic chlorinated solvents to minimize environmental impact.
Scale‑up of reductive amination steps frequently employs hydrogenation over Raney nickel or ruthenium catalysts under pressurized hydrogen atmosphere. The choice of catalyst and ligand is critical for controlling regioselectivity and minimizing side reactions such as over‑reduction or polymerization. In addition, the purification of large‑scale batches often relies on recrystallization or chromatographic techniques such as flash chromatography, reverse‑phase high‑performance liquid chromatography (HPLC), or simulated moving bed (SMB) chromatography for continuous purification.
When the final product is intended for pharmaceutical use, Good Manufacturing Practice (GMP) guidelines dictate rigorous quality control procedures. Analytical methods such as HPLC, mass spectrometry, and nuclear magnetic resonance are employed to confirm purity, stereochemistry, and identity of the product. In addition, residual solvent analysis, heavy metal testing, and assay determination are required to meet regulatory standards.
Applications and Uses
Pharmaceutical Applications
Compounds with the C28H29NO formula are frequently encountered as intermediates or final products in the synthesis of therapeutic agents targeting central nervous system disorders, oncology, and infectious diseases. The presence of an amide linkage combined with aromatic heterocycles confers the ability to engage in π–π stacking interactions with protein targets such as enzymes or receptors. For instance, analogues of the dopamine D2 receptor antagonist may feature a bis(aryl) amide core, where the N‑methyl or N‑alkyl substituent modulates binding affinity.
In oncology, C28H29NO compounds can serve as prodrugs or inhibitors of specific kinases. By incorporating a methoxy substituent at a strategic position, the metabolic stability of the molecule can be enhanced, leading to improved pharmacokinetic profiles. Additionally, the indole scaffold is a privileged structure in medicinal chemistry, often yielding potent inhibitors of topoisomerase or microtubule dynamics.
Antimicrobial and antiviral agents may also arise from this molecular formula, particularly when the nitrogen atom participates in a heterocyclic system that mimics natural nucleosides or amino acids. The design of such molecules involves tailoring electronic properties to disrupt viral replication or bacterial cell wall synthesis.
Material Science
In the realm of organic electronics, C28H29NO compounds are employed as building blocks for organic light‑emitting diodes (OLEDs), organic photovoltaic (OPV) devices, and field‑effect transistors (OFETs). The extended conjugated system provides efficient charge transport and light absorption characteristics. For example, a bis(aryl) amide core conjugated with a pyridine ring can serve as an electron‑transporting material in OLED cathodes, while the presence of a methoxy group enhances solubility in organic solvents, facilitating solution processing.
Fluorescent dyes derived from indole or benzamide structures find applications in bioimaging and sensor technologies. The tunable emission wavelengths, ranging from blue to green, are achieved by adjusting the substitution pattern on the aromatic rings. These dyes can be incorporated into polymer matrices or immobilized on surfaces for detecting analytes such as metal ions or small biomolecules.
Furthermore, C28H29NO compounds can act as ligands in coordination polymers or metal‑organic frameworks (MOFs), where the nitrogen atom coordinates to metal centers such as zinc, copper, or lanthanides. The resulting porous structures are explored for gas storage, catalysis, and separation processes.
Other Uses
Biochemical research frequently employs C28H29NO compounds as probe molecules to study enzyme kinetics, receptor binding, or signal transduction pathways. Radiolabeled analogues of these molecules allow for positron emission tomography (PET) imaging of brain activity or tumor metabolism. Additionally, they serve as standards in analytical chemistry, particularly in the validation of chromatographic methods and spectroscopic techniques.
In agrochemical development, such compounds may function as herbicides or growth regulators, where the amide linkage and aromatic heterocycles interact with plant metabolic enzymes. The careful placement of functional groups ensures selectivity and reduced phytotoxicity.
Future Directions
Research into C28H29NO compounds continues to evolve, with an emphasis on developing more selective and potent therapeutic agents while simultaneously improving environmental sustainability. Computational drug design methods such as quantitative structure–activity relationship (QSAR) modeling, molecular docking, and machine‑learning algorithms aid in predicting binding affinity and metabolic stability, expediting the discovery process.
In materials chemistry, emerging techniques such as nanostructured film deposition, inkjet printing, and 3D printing of organic semiconductors rely on high‑solubility analogues derived from the C28H29NO scaffold. Enhancing mechanical flexibility while maintaining electronic performance is a key challenge, often addressed by incorporating flexible aliphatic linkers or heteroatom‑rich motifs.
Finally, the integration of green chemistry principles - such as the use of bio‑derived solvents, recyclable catalysts, and energy‑efficient reaction conditions - remains paramount. The pursuit of lower carbon footprints and reduced waste generation aligns with the broader goals of sustainable chemistry, ensuring that future generations benefit from the advances enabled by these versatile compounds.
Conclusion
In summary, the molecular formula C28H29NO encapsulates a versatile class of compounds that exhibit a range of physicochemical properties conducive to applications in pharmaceuticals and advanced materials. Their high thermal stability, moderate polarity, and spectroscopic fingerprints facilitate synthesis, characterization, and deployment in cutting‑edge technologies. Ongoing research strives to refine synthetic routes, enhance biological activity, and promote sustainable practices, thereby expanding the utility of these molecules across diverse scientific disciplines.





No comments yet. Be the first to comment!