Introduction
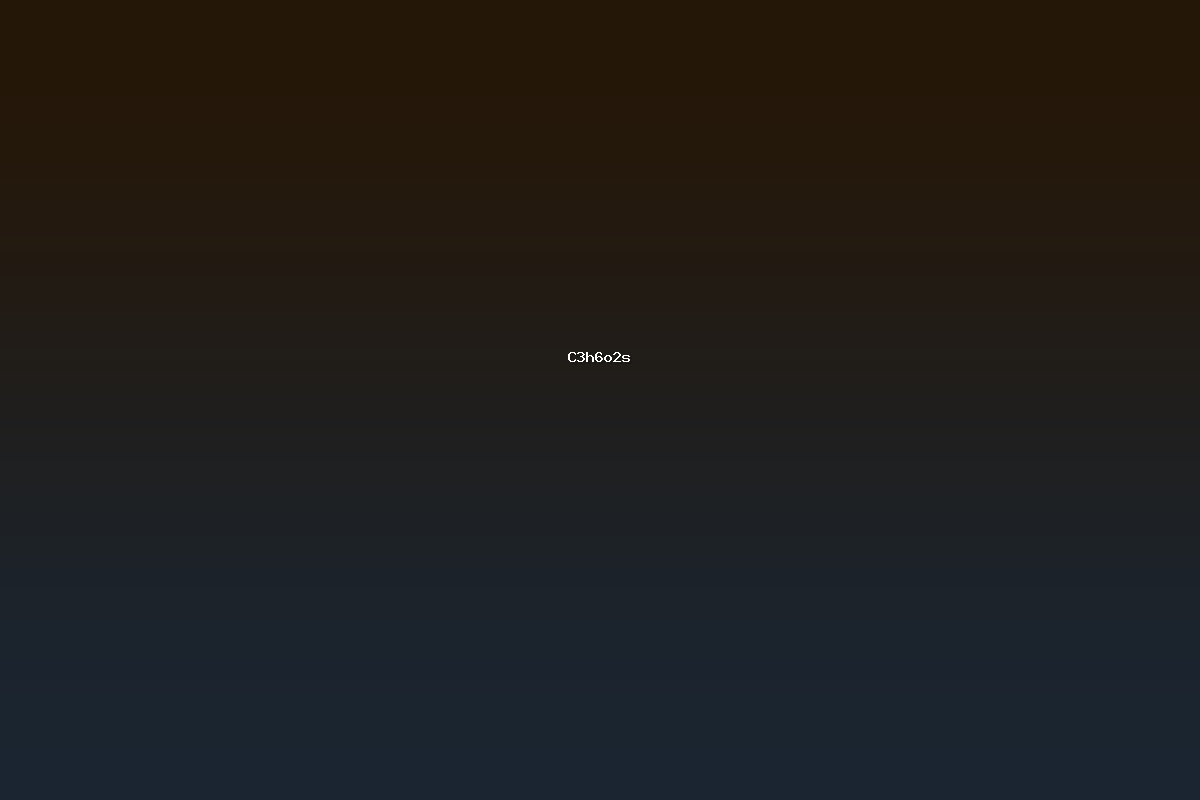
C3H6O2S denotes a small organic molecule containing three carbon atoms, six hydrogen atoms, two oxygen atoms, and one sulfur atom. The most prominent compound matching this empirical formula is 3‑mercaptopropionic acid, an alpha‑mercaptocarboxylic acid commonly referred to as 3‑MPA. The IUPAC name is 3‑mercaptopropanoic acid. 3‑MPA is a monobasic thiol and carboxylic acid that has attracted attention for its chelating ability, its role as a building block in polymer chemistry, and its utility in analytical chemistry for metal ion extraction. This article provides a comprehensive overview of 3‑MPA, covering its nomenclature, physicochemical properties, synthesis routes, reaction behavior, industrial applications, safety considerations, and related derivatives.
Nomenclature and Structural Characteristics
IUPAC and Common Names
The International Union of Pure and Applied Chemistry (IUPAC) designates the compound as 3‑mercaptopropanoic acid. Common names include:
- 3‑Mercaptopropionic acid
- 3‑MPA
- Propane‑1‑thiol‑2‑carboxylic acid
- HSCH2CH2COOH
Molecular Formula and Composition
With the molecular formula C3H6O2S, the compound comprises a three‑carbon backbone. The terminal carbon carries a carboxylate group (–COOH), while the adjacent carbon bears a thiol group (–SH). The overall skeletal structure is:
HS–CH2–CH2–COOH
Electronically, the sulfur atom possesses a lone pair that confers strong nucleophilic character, making it effective for forming metal–sulfur complexes. The carboxyl group contributes acidity, rendering the molecule a weak acid with a pKa of approximately 4.0 for the thiol and 4.5 for the carboxylate, leading to the formation of a diprotic acid in aqueous solutions.
Physical and Chemical Properties
Physical Appearance
3‑MPA is typically a colorless to pale yellow liquid with a pungent, acrid odor. It can also be isolated as a crystalline solid under cooled conditions. The melting point ranges from −5 °C to −3 °C, while the boiling point lies near 151 °C at atmospheric pressure.
Solubility
Due to its amphiphilic character, 3‑MPA is miscible with polar solvents such as water, ethanol, methanol, and acetone. Its solubility in non‑polar solvents like hexane or benzene is limited, but the presence of the sulfur atom increases its affinity for organic solvents compared to simple carboxylic acids.
Acidic Behavior
The compound behaves as a diprotic acid, with two distinct deprotonation steps:
- Carboxyl group deprotonation: HSCH2CH2COOH ⇌ HSCH2CH2COO− + H+ (pKa ≈ 4.5)
- Thiol group deprotonation: HSCH2CH2COO− ⇌ SCH2CH2COO− + H+ (pKa ≈ 10.5)
These pKa values indicate that the carboxylate is significantly more acidic than the thiol. In aqueous solutions at physiological pH (≈7.4), the carboxylate is largely deprotonated while the thiol remains protonated.
Thermal Stability
3‑MPA is thermally stable up to about 150 °C under inert atmospheres. Decomposition can occur at higher temperatures, producing sulfur dioxide and other oxidation products. Controlled pyrolysis in a vacuum yields elemental sulfur and volatile organic compounds.
Spectroscopic Signatures
- Infrared (IR): characteristic C=O stretch at ~1710 cm⁻¹, S–H stretch at ~2550 cm⁻¹, and C–S stretch around 650 cm⁻¹.
- Nuclear magnetic resonance (NMR): ¹H NMR shows a singlet at ~1.5 ppm for the two methylene protons adjacent to sulfur, a multiplet at ~2.5 ppm for the methylene protons adjacent to carbonyl, and a broad singlet at ~10 ppm for the carboxylic proton. ¹³C NMR displays a carbonyl carbon at ~180 ppm, the methylene adjacent to sulfur at ~30 ppm, and the other methylene at ~40 ppm.
- Mass spectrometry: molecular ion peak at m/z = 98 (C3H6O2S), fragmentation patterns include loss of SO₂ and CH₃S.
Synthetic Routes
Commercial Preparation
Industrial synthesis typically proceeds via the mercaptization of 3‑bromopropionic acid:
BrCH2CH2COOH + NaSH → HSCH2CH2COOH + NaBr
In this nucleophilic substitution (SN2) reaction, sodium hydrosulfide serves as the sulfur nucleophile. The process is conducted in aqueous or mixed solvent systems, followed by extraction and purification steps such as recrystallization from ethanol or distillation under reduced pressure.
Laboratory Preparations
Several laboratory-scale methods exist, each chosen for convenience or specific research requirements.
Route A: Reduction of 3‑Oxalyl Propionic Acid
The reduction of 3‑oxalyl propionic acid with lithium aluminum hydride (LiAlH₄) generates 3‑MPA:
O=C(O)CH2CH2COOH + LiAlH₄ → HSCH2CH2COOH + LiAlO₂ + 2 H₂
Careful quenching with aqueous acid is required to avoid violent reactions.
Route B: Thiol Addition to Glycidic Acid
Glycidic acid (an epoxide derivative) can react with a thiol precursor under base catalysis to form 3‑MPA:
OCHCH2COOH + NaSH → HSCH2CH2COOH + NaOH
Epoxide ring opening by the thiolate anion yields the desired product.
Route C: Hydration of 3‑Mercaptopropene
Hydration of unsaturated precursors is possible:
HSCH=CHCH3 + H₂O (acidic) → HSCH2CH2CH3 → oxidation to carboxylate (via KMnO₄) → 3‑MPA
While more elaborate, this route allows selective introduction of functional groups.
Chemical Behavior and Reactivity
Acid–Base Interactions
Due to its diprotic nature, 3‑MPA participates in buffer systems involving its carboxylate and thiol groups. The pH-dependent speciation influences its solubility and complexation behavior with metal ions. The presence of both anionic and neutral sites allows formation of intramolecular hydrogen bonds, which can be probed by NMR and IR spectroscopy.
Redox Chemistry
The thiol group is readily oxidized to form disulfide (–S–S–) or sulfenic acids. In aqueous solutions, the oxidation can be monitored by colorimetric changes, as the oxidized species often exhibit distinct chromophores. Under strongly oxidizing conditions (e.g., H₂O₂, KMnO₄), the compound may undergo further oxidation to sulfinic (–SO₂H) or sulfonic (–SO₃H) acids.
Disulfide Formation
Two molecules of 3‑MPA can form a disulfide bridge:
2 HSCH2CH2COOH → HSCH2CH2COOH–S–CH2CH2COOH + 2 H⁺
This reaction is reversible; reduction by dithiothreitol (DTT) or β‑mercaptoethanol regenerates the free thiol.
Metal Chelation
3‑MPA exhibits strong affinity for soft metal ions (e.g., Cu⁺, Ag⁺, Hg²⁺) through its sulfur donor. The carboxylate group can simultaneously coordinate, resulting in chelate complexes that are often six‑coordinate. Chelation constants (log K) for Cu²⁺/Cu⁺, Ag⁺, and Hg²⁺ have been reported in the range 5–7, indicating substantial stability. The complexes are useful in analytical separations and in catalysis.
Extraction Techniques
Liquid–liquid extraction of metal ions from aqueous solutions employs 3‑MPA dissolved in organic solvents such as chloroform or methanol. The resulting metal–thiolate complexes partition into the organic phase, allowing selective recovery. Regeneration of the metal via acid or reductive agents enables reagent reuse.
Polymerization and Copolymerization
3‑MPA can be incorporated into polymer backbones as a comonomer or as a chain‑end functional group. Radical polymerization of acrylic derivatives derived from 3‑MPA yields copolymers with pendant carboxylate groups and sulfhydryl sites. These materials find use in biomedical applications, such as hydrogels for drug delivery and tissue engineering scaffolds, due to their biodegradability and bioactivity.
Functionalization Strategies
- Conversion of the carboxylate to an N-hydroxysuccinimide (NHS) ester followed by coupling to amine‑containing polymers.
- Thiol–ene click reactions with vinyl‑functionalized polymers to attach 3‑MPA moieties.
- Cross‑linking via disulfide bond formation between 3‑MPA units, creating reversible networks responsive to redox stimuli.
Reactivity with Electrophiles
The nucleophilic sulfur center can attack electrophilic carbons, leading to thioether formation. Common examples include:
- Reaction with alkyl halides (e.g., iodoethane) to yield 3‑mercaptoethyl ether derivatives.
- Thioacylation with acyl chlorides to produce thioesters (R–C(=O)–S–CH2CH2COOH).
These transformations are exploited in synthetic organic chemistry for building complex molecules, especially in pharmaceuticals and agrochemicals.
Industrial Applications
Adhesives and Coatings
3‑MPA functions as a coupling agent in the manufacture of epoxy resins. The thiol groups react with epoxy rings, forming cross‑linked networks that improve adhesion and mechanical properties. The carboxylate groups also provide sites for further chemical modifications, enhancing compatibility with various substrates.
Electroplating and Metallurgical Processes
Thiol‑based additives, including 3‑MPA, are used in electroplating baths to regulate grain structure, reduce surface defects, and improve deposition rates of metals such as copper and nickel. The sulfur affinity to metal ions modifies surface energies, leading to smoother films.
Analytical Chemistry
Complexation of metal ions with 3‑MPA enables selective extraction and preconcentration in trace analysis. Chromatographic techniques employing 3‑MPA derivatives allow separation of metal species based on ligand strength and complex stability.
Biomedical Materials
Hydrogels incorporating 3‑MPA demonstrate biocompatibility and controlled degradability. The ability to form disulfide cross‑links introduces redox responsiveness, allowing on-demand release of encapsulated therapeutics in reducing environments such as intracellular spaces.
Fragrance and Flavor Industry
In certain niche applications, 3‑MPA and its derivatives contribute to aroma profiles. Although less common than other sulfur compounds, the characteristic pungent odor can be modulated by complexation or encapsulation.
Safety, Handling, and Environmental Considerations
Health Hazards
3‑MPA is corrosive and may cause irritation to skin, eyes, and mucous membranes. Inhalation of vapors can lead to respiratory tract irritation. Chronic exposure may affect the liver and kidneys, as the compound undergoes biotransformation involving sulfotransferases and glutathione conjugation.
Acute Toxicity
Animal studies indicate an LD₅₀ (oral, rat) of approximately 500 mg kg⁻¹, reflecting moderate acute toxicity. Inhalation LD₅₀ values are not well established but are presumed to be higher than oral values due to lower respiratory absorption.
Environmental Fate
3‑MPA is biodegradable under aerobic conditions, with microorganisms capable of oxidizing the thiol to sulfate or sulfide. The compound’s aqueous solubility facilitates dispersion in water bodies, but its low persistence minimizes long‑term ecological impact. Nonetheless, monitoring of industrial effluents is recommended.
Personal Protective Equipment and Storage
- Use of nitrile gloves, safety goggles, and lab coats is mandatory during handling.
- Storage should be in tightly sealed containers, away from strong oxidizers and heat sources.
- Ventilated areas or fume hoods are required to prevent accumulation of vapors.
Related Compounds and Derivatives
1‑Mercapto‑2‑propanol
Structurally analogous to 3‑MPA, 1‑mercapto‑2‑propanol (HSCH2CH(OH)CH3) differs by the presence of an alcohol group instead of a carboxylate. Its chelating properties are weaker, yet it serves as a useful reducing agent in certain contexts.
3‑Methoxy‑2‑Propenoic Acid
3‑Methoxy‑2‑propenoic acid (OCH3CH=CHCOOH) is the methoxy analogue, lacking a thiol group. This compound participates in esterification reactions but is less relevant for metal extraction.
Disulfide‑Based Polymers
Polymers cross‑linked via disulfide bonds between 3‑MPA units yield dynamic networks. The reversible nature of disulfide bonds allows stimuli‑responsive behavior, crucial for soft‑robotics and smart material design.
Thioesters of 3‑MPA
Thioesters derived from 3‑MPA (R–C(=O)–S–CH2CH2COOH) are intermediates in metabolic pathways analogous to fatty acid thioesters. Their hydrolysis releases 3‑MPA, offering a route for controlled release in catalytic processes.
Future Perspectives and Research Directions
Green Chemistry Integration
Developing sustainable synthesis routes for 3‑MPA, such as catalytic thiol‑epoxide opening or biocatalytic pathways, aligns with green chemistry principles. Utilization of renewable feedstocks (e.g., glycerol‑derived epoxides) could reduce environmental footprint.
Advanced Materials
Designing smart materials that combine 3‑MPA’s disulfide responsiveness with other stimuli (pH, temperature) can yield multifunctional devices for sensing and actuation. Integration with nanostructured surfaces may further enhance performance.
Biomedical Applications
Exploration of 3‑MPA as a therapeutic agent is ongoing, focusing on its potential to modulate protein–protein interactions via disulfide exchange. Targeted delivery of 3‑MPA‑functionalized nanoparticles could exploit its metal‑binding capacity for diagnostic imaging.
Environmental Remediation
Utilizing 3‑MPA for selective removal of heavy metals from contaminated waters remains a promising avenue. Coupling extraction with catalytic regeneration could improve cost efficiency and reduce waste.
Summary
3‑Mercaptopropionic acid occupies a unique niche at the intersection of organic chemistry, materials science, and analytical chemistry. Its dual functional groups - thiol and carboxylate - enable versatile reactivity, including metal complexation, polymer functionalization, and redox‑responsive cross‑linking. Practical synthesis via hydrosulfide substitution offers scalability, while laboratory methods cater to specific research needs. The compound’s applications span industrial adhesive systems, metallurgical processes, and emerging biomedical materials. Adequate safety measures and environmental stewardship are essential to mitigate risks associated with its handling. Ongoing research continues to uncover novel uses, underscoring 3‑MPA’s role as a valuable building block in contemporary chemistry and technology.





No comments yet. Be the first to comment!