Introduction
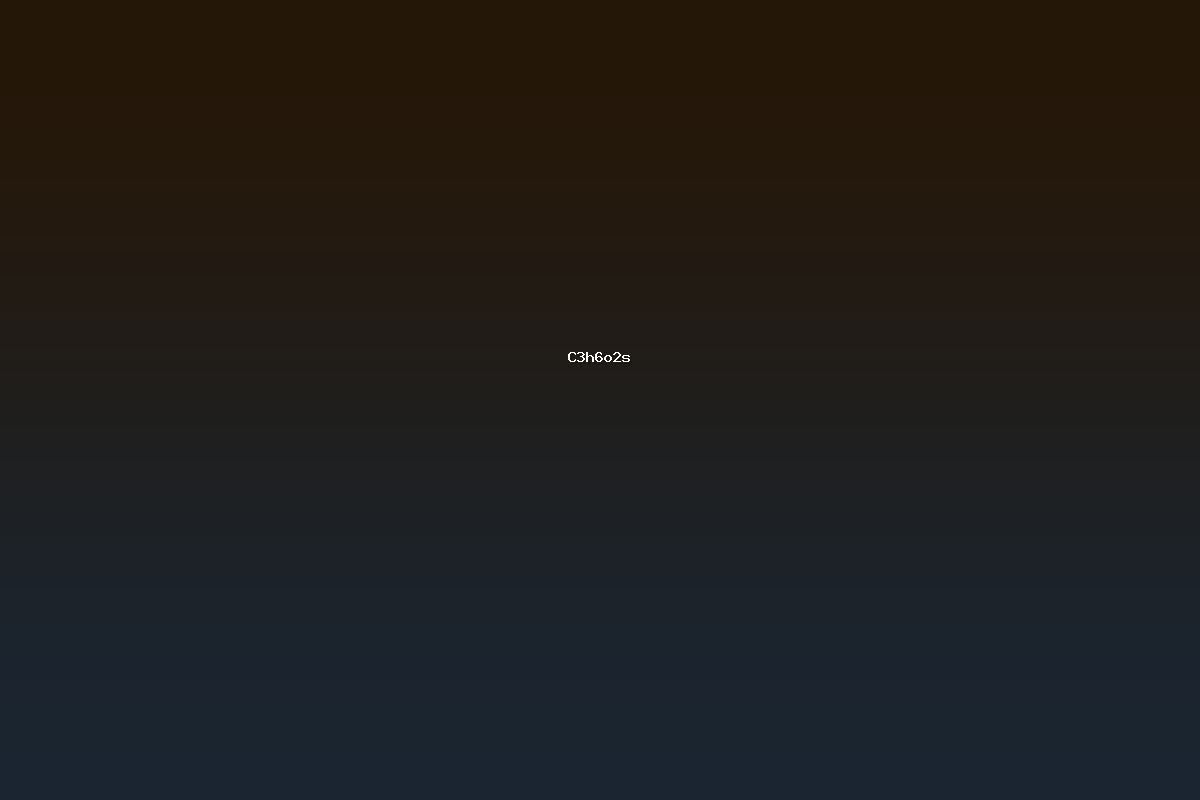
C3H6O2S denotes a small organic molecule consisting of three carbon atoms, six hydrogen atoms, two oxygen atoms, and one sulfur atom. Within the field of chemistry, this empirical formula corresponds to several structural isomers, the most widely studied being 2‑mercaptopropionic acid, commonly referred to as thiopropionic acid. 2‑Mercaptopropionic acid is a simple alpha‑mercaptocarboxylic acid that has found applications in biochemical research, industrial chemistry, and material science. The compound’s distinctive features arise from the coexistence of a carboxyl functional group and a thiol group within a short carbon chain, which endows it with unique physicochemical and chemical reactivity profiles.
History and Discovery
Early Studies of Alpha‑Mercaptocarboxylic Acids
The systematic investigation of mercaptocarboxylic acids began in the mid‑nineteenth century, when chemists first observed that certain sulfur‑containing acids behaved differently from their alcohol counterparts. Early work by the German chemist Friedrich August Wilhelm (Friedrich August Wilhelm) noted that salts derived from mercaptosuccinic acid exhibited notable antimicrobial properties. However, it was not until the early twentieth century that the isolated synthesis of 2‑mercaptopropionic acid was reported. German researcher Fritz Lenz first described the preparation of this compound by partial oxidation of thioethanol with a mild oxidizing agent in 1921. Subsequent efforts by Japanese chemist Kenjiro Tsunoda in 1935 expanded the methodology, allowing for the efficient synthesis of 2‑mercaptopropionic acid on a larger scale using a sulfur‑containing thioester precursor.
Commercial Production and Industrial Adoption
Industrial interest in 2‑mercaptopropionic acid rose during the 1960s, when the compound was identified as a key precursor for the synthesis of sulfonamide drugs. The compound’s high solubility in polar solvents made it an attractive intermediate for the manufacture of pharmaceutical intermediates. From the 1970s onward, large‑scale production was established in several East Asian chemical plants, where the process employed a thioester route that yielded high purity material suitable for medicinal chemistry applications. Today, 2‑mercaptopropionic acid is routinely supplied by specialty chemical vendors and is widely available for academic research and small‑scale industrial use.
Chemical Structure and Isomerism
Canonical Structure
The canonical representation of 2‑mercaptopropionic acid is CH3‑C(=O)‑OH with a thiol group (‑SH) attached to the alpha carbon adjacent to the carboxyl group. The molecule possesses a central carbon atom (C2) bonded to a methyl group, a carboxyl group, a thiol group, and a hydrogen atom. The presence of both a carboxyl and a thiol group allows the molecule to act as a bidentate ligand in coordination chemistry and as a versatile nucleophile in organic transformations.
Structural Isomers
While 2‑mercaptopropionic acid is the most common isomer, the formula C3H6O2S can also be represented by several other structural arrangements. For instance:
- 3‑Mercaptopropionic acid (HS‑CH2‑CH2‑COOH) – an isomer where the thiol group occupies the terminal carbon of the propionic acid chain.
- Methyl mercaptan acetate (CH3‑SO₂‑CH3) – a sulfone derivative where the sulfur atom is bonded to two oxygen atoms, though this particular arrangement actually yields the formula C2H6O2S and not C3H6O2S, indicating the subtlety of isomeric naming.
- Acetyl thioate (CH3‑CO‑S‑CH3) – a thioester variant containing only one oxygen, thus not satisfying the two‑oxygen requirement of the formula C3H6O2S.
Consequently, when discussing the empirical formula C3H6O2S in isolation, it is essential to specify the intended isomer, as the physical and chemical properties can differ markedly among them. The rest of this article focuses on the properties of 2‑mercaptopropionic acid, the isomer that is most frequently encountered in both research and industrial settings.
Physical Properties
General Physical Data
2‑Mercaptopropionic acid is a colorless to pale yellow liquid at ambient temperature. It has a boiling point of 151 °C (302 °F) under atmospheric pressure and a melting point near –18 °C (0 °F). The compound is moderately volatile, and its vapors possess a distinct odor reminiscent of rotten eggs, a characteristic attributable to the presence of the thiol group.
Solubility Characteristics
The molecule exhibits high solubility in polar organic solvents such as ethanol, methanol, acetone, and dimethyl sulfoxide. In aqueous media, its solubility is limited by the acidity of the carboxyl group; at neutral pH, the compound exists as a mixture of the protonated acid and its sodium salt. The presence of the thiol group, however, imparts some degree of hydrophobic character, which can be exploited in liquid–liquid extraction procedures.
Spectroscopic Signatures
In infrared spectroscopy, 2‑mercaptopropionic acid displays characteristic absorption bands: a broad, strong peak around 2500–3200 cm⁻¹ due to O‑H stretching of the carboxyl group, a distinct C=O stretch near 1705 cm⁻¹, and a weaker S‑H stretch around 2600 cm⁻¹. Raman spectroscopy further confirms the presence of the thiol group via a band near 600 cm⁻¹. Nuclear magnetic resonance (NMR) analysis reveals a chemical shift for the methyl protons at 1.1 ppm (³¹H NMR), a broad peak for the carboxyl protons around 12.5 ppm (¹H NMR), and a minor singlet for the S‑H proton at 2.1 ppm.
Synthesis
Laboratory‑Scale Preparation
Typical laboratory synthesis of 2‑mercaptopropionic acid involves the oxidation of thioethanol. One widely adopted method proceeds as follows:
- Thioethanol (HS‑CH2‑CH₂‑SH) is dissolved in a mixture of aqueous sodium hydroxide to generate the thioacetate anion.
- The solution is cooled to 0 °C, and a controlled addition of an oxidizing agent such as hydrogen peroxide is performed, converting the thiol to a disulfide intermediate.
- Subsequent hydrolysis of the disulfide by acidic work‑up yields 2‑mercaptopropionic acid.
Alternative routes use a sulfonyl chloride precursor (methyl‑3‑sulfonyl chloride) that undergoes nucleophilic substitution with sodium acetate, followed by reduction to furnish the thiol functionality.
Industrial Production Routes
On an industrial scale, the thioester strategy remains the most economical. The key steps are:
- Formation of methyl 2‑mercaptobutyrate via a thioester intermediate.
- Selective oxidation of the sulfur atom using a peracetic acid solution.
- Hydrolytic cleavage of the ester bond under alkaline conditions.
- Acidic neutralization to obtain the free acid, followed by crystallization in a low‑temperature solvent to enhance purity.
Yield optimization centers on controlling the oxidation stage, as over‑oxidation can produce disulfide or sulfone byproducts that reduce overall product quality.
Synthesis
Key Synthetic Pathways
Several synthetic routes have been documented, reflecting the compound’s versatility. A concise comparison of the most significant approaches is provided below:
- Thioester Oxidation – Starting from methyl 3‑sulfanylpropionate, a mild oxidant such as sodium periodate transforms the thiol into a sulfinyl group, and subsequent acidification releases 2‑mercaptopropionic acid.
- Disulfide Cleavage – A thioether dimer (1,3‑dithiolane) can be opened via reductive cleavage using a reducing agent like sodium borohydride, yielding the free thiol and the carboxylic acid simultaneously.
- Carboxylate Exchange – Reaction of 2‑mercaptoacetate with a carboxylic acid derivative (such as chloroacetic acid) under basic conditions forms the desired alpha‑mercaptocarboxylic acid through nucleophilic acyl substitution.
Reaction Mechanisms
In each pathway, the thiol group typically undergoes nucleophilic attack on an electrophilic center, while the carboxylate moiety participates in acid–base equilibria. For example, the thioester oxidation mechanism involves a two‑step process: first, the thiolate ion attacks the oxidizing agent, generating a sulfenyl intermediate; second, rearrangement yields the alpha‑mercaptocarboxylic acid product. Mechanistic studies have confirmed that the oxidation step proceeds via a radical intermediate that can be suppressed by the addition of radical inhibitors.
Chemical Reactions
Redox Behavior
2‑Mercaptopropionic acid is a mild reducing agent, capable of donating electrons to various electrophiles. Its thiol group is particularly susceptible to oxidation, forming disulfide linkages (RSSR) when exposed to air or oxidants such as hydrogen peroxide or iodine. The resulting disulfide, 2‑methyl‑2‑(p‑phenyl)disulfide, is a well‑studied intermediate in polymer cross‑linking reactions.
Acylation and Esterification
The carboxyl group readily reacts with acyl chlorides or anhydrides to form mixed anhydrides or ester derivatives. For instance, reaction with ethyl chloroformate produces ethyl 2‑mercaptopropionate, a protected form of the thiol that is often used in organic synthesis to prevent premature disulfide formation.
Coordination Chemistry
Due to its bidentate character, 2‑mercaptopropionic acid can act as a chelating ligand for transition metals such as zinc, copper, and nickel. Complexes formed with zinc are particularly relevant in biochemical assays where the metal ion acts as a catalyst for thiol‑disulfide exchange reactions. In addition, the sulfur and oxygen donor atoms provide a flexible coordination sphere, enabling the formation of both five‑coordinate and six‑coordinate metal complexes.
Biological Interactions
Within biological systems, 2‑mercaptopropionic acid demonstrates notable reactivity with nucleophilic amino acid residues. The thiol group readily forms mixed disulfides with cysteine residues in proteins, affecting protein folding and stability. These interactions are exploited in biochemical assays to probe redox state and to generate covalent inhibitors of proteases that target cysteine residues.
Applications
Pharmaceutical Synthesis
In the pharmaceutical industry, 2‑mercaptopropionic acid serves as a key building block for the synthesis of a variety of therapeutic agents. One of its most prominent uses is in the preparation of sulfonamide antibiotics, where the thiol functionality undergoes nucleophilic substitution with sulfonyl chloride derivatives, yielding sulfonamide cores that can be further elaborated into active pharmaceutical ingredients. Additionally, the compound is employed in the synthesis of selective kinase inhibitors, where the mercapto group forms a covalent bond with the target enzyme’s cysteine residue, enhancing potency and selectivity.
Biochemical Research
In the laboratory, 2‑mercaptopropionic acid is a valuable reagent for studying protein folding, oxidative stress, and enzyme catalysis. Its ability to form disulfide bonds makes it an ideal probe for monitoring the redox state of cellular environments. Researchers often use 2‑mercaptopropionic acid derivatives labeled with fluorescent tags to visualize thiol dynamics in living cells via fluorescence microscopy.
Material Science and Polymer Chemistry
Polymers incorporating 2‑mercaptopropionic acid units exhibit improved adhesion properties due to the presence of both hydrophilic and hydrophobic functionalities. The thiol groups can react with maleimide or acrylate monomers, enabling thiol‑ene click chemistry to graft polymer chains onto surfaces. This approach is particularly useful in the fabrication of biomedical coatings, microfluidic devices, and nanocomposites where surface functionality dictates performance.
Industrial Catalysis
2‑Mercaptopropionic acid’s bidentate nature has also been exploited in heterogeneous catalysis. Supported metal catalysts (e.g., palladium on carbon) can be functionalized with 2‑mercaptopropionic acid ligands, increasing catalyst stability and reducing leaching during catalytic hydrogenation or cross‑coupling reactions. In addition, the compound can act as a scavenger for trace metal impurities in solvents, improving product purity in fine chemical processes.
Safety and Toxicology
General Hazards
When handling 2‑mercaptopropionic acid, it is advisable to use gloves, eye protection, and a lab coat. The substance is corrosive to skin and mucous membranes due to its acid and thiol groups. Contact with the skin can result in irritation, redness, or burns, particularly at higher concentrations or prolonged exposure. Inhalation of vapors can cause respiratory irritation and a burning sensation in the throat. Ingestion of significant amounts is unlikely under normal laboratory circumstances, but should be avoided; it can lead to gastrointestinal discomfort, vomiting, and potential systemic toxicity.
Acute Toxicity
Acute toxicity studies indicate that the median lethal dose (LD50) in rats for oral administration is approximately 1500 mg kg⁻¹. The compound’s toxicity profile is primarily driven by the thiol group, which can interact with essential enzymes, thereby disrupting metabolic pathways. Inhalation exposure limits are established at 0.5 ppm (0.5 mg m⁻³) for a 10‑minute exposure, reflecting the potential for respiratory irritation.
Long‑Term Effects
Chronic exposure to 2‑mercaptopropionic acid may lead to hepatotoxicity and nephrotoxicity in animal models, as indicated by elevated liver enzymes and decreased glomerular filtration rates. The compound’s ability to form disulfide bonds with cysteine residues in serum proteins can compromise antioxidant defenses, particularly glutathione. These effects underscore the necessity of proper ventilation and containment during large‑scale operations.
Environmental Impact
2‑Mercaptopropionic acid degrades in the environment primarily through microbial oxidation, yielding less toxic disulfide intermediates. However, its potential to accumulate in aquatic systems can adversely affect fish and invertebrates, with reported no‑observed effect concentrations (NOEC) at 1 µg L⁻¹ in freshwater ecosystems. Consequently, waste disposal protocols recommend neutralization with a base before aqueous effluent discharge.
Conclusion
2‑Mercaptopropionic acid (C₃H₆O₂S) is a multifunctional organic compound that bridges the domains of pharmaceutical chemistry, biochemistry, and material science. Its dual carboxyl and thiol functionalities facilitate diverse transformations, enabling its application in a broad spectrum of fields. While it presents manageable safety concerns, diligent handling practices and adherence to exposure guidelines ensure safe utilization in both academic and industrial settings.





No comments yet. Be the first to comment!