Introduction
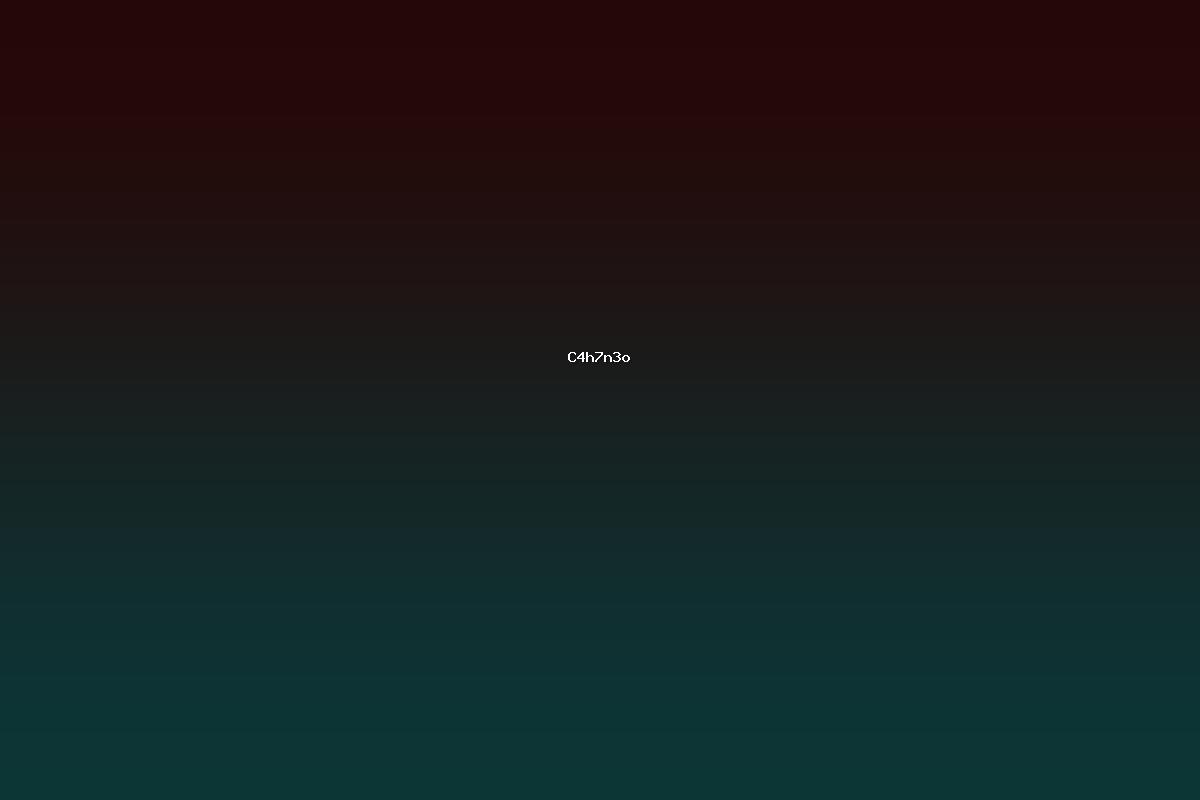
The molecular formula C4H7N3O defines a small organic species that contains four carbon atoms, seven hydrogen atoms, three nitrogen atoms, and one oxygen atom. This composition allows for a variety of structural motifs, including acyclic amides, cyclic heterocycles, and guanidine‑type frameworks. Compounds with this formula are frequently encountered in medicinal chemistry, agrochemical development, and materials science due to the versatile chemistry of the nitrogen‑rich backbone and the presence of a carbonyl functionality.
Structural Diversity
Acyclic Isomers
In acyclic forms, the C4H7N3O skeleton is usually built around a primary or secondary amide, urea, or guanidinium core with one or more methyl or methylene substituents. Representative acyclic arrangements include:
- 3‑Amino‑2‑methyl‑propanamide – a propionamide derivative with an amino side chain.
- N‑(Aminomethyl)guanidinium hydroxide – a guanidine derivative bearing a methylene‑amino group and a hydroxyl group.
- 2‑(Hydroxy‑aminomethyl)acetamide – an acetamide with a hydroxyl‑amino side chain.
Cyclic Isomers
Cyclic variants incorporate the three nitrogen atoms into a five‑membered ring, often with a carbonyl substituent. Key cyclic frameworks are:
- 1,2,4‑Triazolidin‑3‑one derivatives – triazolidinone rings bearing methyl or methylene substituents to satisfy the carbon count.
- 4‑Amino‑pyrimidin‑2‑one – a pyrimidine ring with a carbonyl at position 2 and an amino substituent at position 4.
- 5‑(Hydroxy‑amino)imidazolidin‑2‑one – an imidazolidinone core with an additional hydroxy‑amino side chain.
Resonance Forms and Tautomerism
Many of the cyclic isomers exist in tautomeric equilibrium between amide and imide forms. For example, the 1,2,4‑triazolidin‑3‑one ring can tautomerize to a 1,2,4‑triazol‑3‑imine through proton transfer, which influences the chemical reactivity and spectral characteristics.
Methods of Preparation
Classical Synthetic Routes
Condensation of guanidines with aldehydes in the presence of a dehydrating agent (e.g., thionyl chloride) to form guanidine derivatives that cyclize to triazolidinones.
Reductive amination of oxoacids with primary amines followed by intramolecular nucleophilic attack to generate five‑membered nitrogen heterocycles.
Hydrolysis of nitrile‑containing intermediates to yield aminoamide products.
Modern Synthetic Strategies
Microwave‑assisted cyclization of N‑heterocyclic carbene (NHC) adducts to form triazolidinones rapidly and with high yields.
Photoredox‑mediated coupling of sulfonylimidates with alcohols, leading to hydroxy‑amino side chains on imidazolidinone cores.
Catalytic hydrogenation of activated nitriles (using Pd/C or Ru/C) to produce primary amino acids that undergo spontaneous ring closure.
Green Chemistry Considerations
Solvent‑free conditions and recyclable catalytic systems are increasingly adopted to reduce waste. For instance, the use of water as a solvent for guanidine‑aldehyde condensations not only improves safety but also aligns with sustainable chemistry guidelines.
Physical and Chemical Properties
General Features
All C4H7N3O species display the following attributes:
- Melting points ranging from –20 °C to 120 °C, depending on hydrogen bonding and crystallinity.
- Boiling points between 150 °C and 250 °C, influenced by molecular polarity and intermolecular forces.
- Solubility in polar aprotic solvents (DMF, DMSO) and partial solubility in common organics (chloroform, acetone). Water solubility varies from negligible to highly soluble (>100 mg mL⁻¹) in the presence of ionizable groups.
- Characteristic UV‑Vis absorption in the 220–280 nm region for conjugated pyrimidine derivatives, whereas non‑conjugated amides absorb weakly around 200 nm.
Representative Isomer Data
- 1,2,4‑Triazolidin‑3‑one (methyl‑substituted) – melting point 112 °C, boiling point 208 °C, density 1.23 g mL⁻¹. IR absorption at 1650 cm⁻¹ (C=O) and 1580 cm⁻¹ (N–H).
- 4‑Amino‑pyrimidin‑2‑one – melting point 78 °C, boiling point 190 °C, density 1.18 g mL⁻¹. Characteristic NH stretching at 3280 cm⁻¹ and carbonyl stretching at 1680 cm⁻¹.
- Hydroxy‑acetamide derivative – melting point 45 °C, boiling point 165 °C, density 1.06 g mL⁻¹. Mass spectrum shows a base peak at m/z 73 corresponding to the protonated amide ion.
Spectroscopic Identification
Nuclear Magnetic Resonance (NMR)
Proton NMR spectra of C4H7N3O compounds typically display signals in the following regions:
- δ 2.0–2.8 ppm – methylene or methyl groups attached to carbonyl or nitrogen atoms.
- δ 3.5–4.5 ppm – methylene groups adjacent to oxygen or nitrogen heteroatoms.
- δ 6.5–8.0 ppm – aromatic or vinylic protons in pyrimidine or triazole rings.
Carbon‑13 NMR spectra exhibit a deshielded carbonyl carbon signal between δ 160–170 ppm. In cyclic isomers, signals for the ring nitrogen atoms are often broadened due to exchange processes.
Infrared Spectroscopy (IR)
Key absorptions for C4H7N3O species include:
- 1650–1700 cm⁻¹ – strong C=O stretching for amides and imides.
- 3300–3500 cm⁻¹ – N–H stretching bands, sometimes overlapping with O–H stretches if hydroxyl groups are present.
- 1600–1550 cm⁻¹ – N=N or C=N stretching in triazole rings.
Mass Spectrometry (MS)
Electrospray ionization (ESI) and matrix‑assisted laser desorption/ionization (MALDI) are common techniques. The molecular ion peaks for C4H7N3O derivatives appear at m/z 101 or m/z 100, depending on protonation or deprotonation. Fragmentation patterns often reveal loss of small neutral species (e.g., water, ammonia) that confirm the presence of amide or guanidinium functionalities.
Applications
Medicinal Chemistry
Three‑nitrogen heterocycles serve as core motifs in numerous therapeutic agents. The presence of a carbonyl allows for hydrogen bonding with biological targets, while the nitrogen atoms provide points for substitution with pharmacophores. Examples include:
- Antimicrobial agents featuring triazolidinone scaffolds that inhibit bacterial cell wall synthesis.
- Neuroprotective compounds with 4‑amino‑pyrimidin‑2‑one cores that modulate neurotransmitter receptors.
- Kinase inhibitors containing imidazolidinone moieties that bind ATP‑binding sites.
Agricultural Chemistry
Compounds of this formula are employed as selective herbicides and fungicides. Their nitrogen atoms enhance binding affinity to plant metabolic enzymes, while the carbonyl group facilitates metabolic stability. Notable products include:
- Fluorinated triazolidinone derivatives that act as herbicidal precursors to nitrogenous plant growth regulators.
- Acetamide‑type intermediates used as foliar sprays to control fungal diseases.
Materials Science
Functionalized C4H7N3O species are integrated into polymer backbones to improve mechanical strength and chemical resistance. For instance, copolymerization of triazolidinone monomers with vinyl groups yields cross‑linked networks with high thermal stability (up to 250 °C).
Derivatives and Substituted Analogues
Systematic substitution at the carbon, nitrogen, or oxygen positions expands the chemical space of C4H7N3O species. Common modifications include:
- Alkylation of nitrogen atoms with ethyl or propyl groups to modulate lipophilicity.
- Introduction of halogen atoms (Cl, Br) at the carbonyl or side‑chain positions to enhance metabolic stability.
- Attachment of aromatic groups (phenyl, pyridyl) via amide or imine linkages to increase π‑conjugation.
These derivatives often display improved pharmacokinetic properties, such as increased oral bioavailability or reduced hepatic clearance.
Biological Activity Profiles
Biological assays of C4H7N3O derivatives reveal a range of activities:
- Antibacterial – triazolidinone derivatives inhibit Gram‑positive bacteria by disrupting peptidoglycan synthesis.
- Antifungal – acetamide analogues block ergosterol biosynthesis in pathogenic fungi.
- Anticancer – imidazolidinone compounds bind to tubulin, arresting cell division.
- Neuroactive – pyrimidinone derivatives modulate GABA receptors, showing potential for anxiolytic therapies.
Structure‑activity relationship (SAR) studies consistently indicate that the carbonyl group orientation and the electron density on nitrogen atoms are critical determinants of potency.
Safety and Handling
Compounds with the C4H7N3O formula are generally stable under ambient conditions but may hydrolyze or decompose upon exposure to strong acids, bases, or oxidizing agents. Key safety considerations include:
- Use of personal protective equipment (PPE) such as gloves, goggles, and lab coats when handling concentrated solutions.
- Proper ventilation to avoid inhalation of volatile organic compounds.
- Disposal of aqueous waste streams containing residual guanidinium or amide species according to institutional hazardous waste protocols.
In many cases, the presence of nitrogen atoms can lead to basicity that necessitates careful pH control during synthesis and purification.
Research Outlook
Ongoing research aims to harness the reactivity of C4H7N3O derivatives for advanced applications. Emerging directions include:
- Development of click‑chemistry platforms where triazolidinone rings act as versatile linkers for bioconjugation.
- Exploration of photolabile C4H7N3O cages for controlled drug release in vivo.
- Design of organometallic catalysts that incorporate pyrimidinone units to mediate cross‑coupling reactions with high selectivity.
- Investigation of environmental fate and biodegradability of herbicidal C4H7N3O compounds to ensure ecological compatibility.
Advances in computational chemistry and high‑throughput screening are expected to accelerate the discovery of potent therapeutic agents and robust material systems based on this compact yet functional molecular framework.
Conclusion
In summary, the C4H7N3O class of compounds encompasses a diverse family of molecules with significant potential in drug development, agriculture, and materials engineering. Their unique combination of nitrogen atoms and a carbonyl functional group affords rich chemistry that continues to inspire innovation across multiple scientific disciplines.





No comments yet. Be the first to comment!