Introduction
C4H7N3O is the empirical formula for a family of organic compounds that contain four carbon atoms, seven hydrogen atoms, three nitrogen atoms, and one oxygen atom. The formula corresponds to a small heterocyclic system that can be represented by various isomeric structures, each possessing distinct physical, chemical, and biological characteristics. The presence of multiple nitrogen atoms in a compact ring framework allows these molecules to participate in a variety of bonding interactions, making them useful as building blocks in medicinal chemistry, agrochemical synthesis, and material science.
Structural Diversity and Isomerism
General Structural Features
All molecules with the formula C4H7N3O share the same molecular weight of 95.10 g mol⁻¹ and a degree of unsaturation of three. This degree of unsaturation indicates that each molecule contains a combination of rings and/or multiple bonds equivalent to three double bond equivalents. Typical structural motifs include:
- A five‑membered heteroaromatic ring containing two nitrogen atoms and one oxygen atom (e.g., 1,2,4‑triazol‑5‑one). This arrangement yields a ring with one carbonyl group and two ring nitrogens.
- A saturated five‑membered heterocycle (imidazolidin‑2‑one) containing two nitrogens and one oxygen, with a carbonyl substituent at the ring position.
- Linear or branched chain amides or amidines that include an amide nitrogen and an additional imine nitrogen.
Substituents such as amino or methyl groups can be added to the core skeleton, leading to an extensive set of isomeric possibilities. For example, 2‑amino‑1,3,4‑triazol‑5‑one, 4‑amino‑1,2,3‑triazol‑5‑one, and 5‑amino‑1,3,4‑triazol‑2‑one are all isomers that fit the C4H7N3O formula but differ in the position of the amino substituent and the placement of the carbonyl group.
Common Isomers
Among the isomers that have been characterized experimentally, the following are the most frequently encountered in the literature:
- 2‑Amino‑1,3,4‑triazol‑5‑one (also known as 5‑amino‑1,3,4‑triazolone). This compound is typically isolated as a crystalline solid and exhibits strong hydrogen‑bonding capabilities through both its amine and carbonyl groups.
- 4‑Amino‑1,2,3‑triazol‑5‑one. The substitution pattern results in a slightly different electron distribution, affecting its basicity and reactivity towards electrophiles.
- 5‑Amino‑1,3,4‑triazol‑2‑one. This isomer is less common but can be synthesized via cyclization of appropriately substituted amidine precursors.
- Imidazolidin‑2‑one derivatives such as N‑methyl‑imidazolidin‑2‑one. These compounds are saturated rings containing two nitrogens and one carbonyl oxygen, and they often serve as chiral auxiliaries or ligands in asymmetric synthesis.
Structural elucidation for each isomer typically involves a combination of NMR spectroscopy, mass spectrometry, and X‑ray crystallography to confirm the placement of heteroatoms and the presence of tautomeric forms.
Physical Properties
General Physical Characteristics
Compounds with the C4H7N3O formula are generally small, volatile liquids or crystalline solids at ambient temperature. Their boiling points range from 90 °C to 120 °C, depending on the degree of conjugation and hydrogen‑bonding capacity. Solubility is influenced by the presence of polar functional groups; most isomers are soluble in polar solvents such as water, ethanol, and dimethyl sulfoxide, while only modest solubility is observed in non‑polar solvents like hexane.
Spectroscopic Signatures
In ^1H NMR spectra, signals for the amine protons typically appear as broad multiplets in the range 3.5–5.5 ppm, whereas the aromatic or heteroaromatic protons resonate between 6.5 and 8.5 ppm. The carbonyl carbon of the amide or lactam group shows a strong absorption in the ^13C NMR spectrum near 160–170 ppm. Infrared spectroscopy reveals a sharp absorption at 1660–1690 cm⁻¹ attributable to the C=O stretching vibration, while N–H bending vibrations appear between 3200 and 3400 cm⁻¹.
Thermal Stability
Thermogravimetric analysis (TGA) indicates that most isomers decompose between 250 °C and 300 °C, releasing nitrogen‑containing fragments such as NH₃, HCN, or N₂. Differential scanning calorimetry (DSC) measurements show exothermic events corresponding to ring opening or decarboxylation in some derivatives, which informs the choice of synthesis and purification conditions.
Chemical Properties
Reactivity of the Carbonyl Group
The carbonyl functionality present in all isomers is an electrophilic center susceptible to nucleophilic attack. In aqueous media, addition of water to the carbonyl results in the reversible formation of geminal diols, particularly in the case of the 2‑amino‑1,3,4‑triazol‑5‑one isomer. The stability of the gem-diol depends on the surrounding heteroatom arrangement; a neighboring nitrogen can stabilize the hydroxyl via hydrogen bonding.
N‑Substitution and Tautomerism
Amine nitrogens in these compounds can undergo protonation, generating positively charged species that are more reactive toward electrophilic substitution. Additionally, tautomeric equilibria are observed between enol and keto forms, especially for the 5‑amino‑1,3,4‑triazol‑2‑one isomer. This tautomerism can be detected by ^1H NMR, where the chemical shift of the amino proton changes depending on the tautomeric state. Protonation constants (pK_a) for the ring nitrogens vary between 2.5 and 6.0, reflecting the electron‑withdrawing influence of the adjacent carbonyl group.
Oxidation and Redox Behavior
Oxidation of the heterocyclic ring typically leads to ring opening and the formation of N‑oxide species. For example, treatment of 2‑amino‑1,3,4‑triazol‑5‑one with peracids yields the corresponding N‑oxide, which can be further converted to a nitroso derivative under reductive conditions. Electrochemical studies using cyclic voltammetry demonstrate a reversible reduction wave at –0.8 V vs. Ag/AgCl, attributed to the reduction of the ring nitrogen atoms to form a radical anion.
Acid–Base Behavior
Due to the presence of multiple basic nitrogens, the molecules can act as diprotic acids or bases depending on the pH. In strongly basic media (pH > 10), deprotonation of the amino group occurs, generating an anionic species with increased nucleophilicity. In acidic environments (pH
Synthesis
General Synthetic Strategies
Preparation of C4H7N3O isomers generally follows one of two major synthetic routes: (1) cyclization of amidine or amidoxime precursors, or (2) reductive amination of appropriate heterocyclic aldehydes and amines.
Cyclization of Amidines
Amidines bearing an α‑hydroxyl or acyl group are heated in the presence of a dehydrating agent such as acetic anhydride or polyphosphoric acid. The reaction proceeds through the formation of an intermediate imine that cyclizes to a lactam, generating the triazolone or imidazolidinone core. The reaction is typically conducted at temperatures between 100 °C and 140 °C, with yields ranging from 45 % to 75 % depending on the substituent pattern.
Reductive Amination
Aldehyde intermediates such as 2‑amino‑1,3,4‑triazole aldehydes are reacted with ammonium salts or primary amines in the presence of a reducing agent (e.g., sodium cyanoborohydride). This approach allows selective formation of amino‑substituted triazolones. The reaction is performed in aqueous or mixed solvent systems at room temperature to minimize over‑reduction or side‑reaction.
Alternative Routes
Some derivatives can be accessed via the Paal–Knorr synthesis, where a 1,4‑dicarbonyl compound reacts with a diamine under acidic conditions to produce a 1,3,4‑triazole ring. Subsequent oxidation or reduction steps introduce the carbonyl functionality, yielding the target C4H7N3O compound. This route is advantageous for large‑scale production due to the use of inexpensive starting materials and mild reaction conditions.
Purification Techniques
After synthesis, the crude product is typically purified by recrystallization from ethanol or by column chromatography using silica gel with a gradient of hexane/ethyl acetate. Thin‑layer chromatography (TLC) monitoring uses a UV detector at 254 nm and a spot reagent of ninhydrin to visualize amine groups. Final purity (> 95 %) is confirmed by high‑performance liquid chromatography (HPLC) coupled with mass spectrometry (MS).
Applications
Medicinal Chemistry
Due to their nitrogen‑rich heterocyclic framework, C4H7N3O isomers have been investigated as pharmacophores in drug discovery. The triazolone core provides a rigid scaffold that can interact with biological targets such as kinases, proteases, and nucleic acid enzymes. Small‑molecule inhibitors containing this moiety have demonstrated activity against a range of pathogens, including bacterial species that produce β‑lactamases. Furthermore, the amine substituent allows for conjugation with polyethylene glycol (PEG) chains, improving solubility and pharmacokinetics.
Fluorogenic Probes
Derivatives of the 5‑amino‑1,3,4‑triazol‑2‑one scaffold have been incorporated into fluorogenic probe systems that detect enzymatic activity. When conjugated to a fluorophore such as fluorescein, the probe exhibits a significant change in fluorescence upon enzymatic cleavage of the amide bond, enabling real‑time monitoring of protease activity in cellular assays.
Agricultural Chemistry
Some isomers have shown pesticidal activity against nematodes and fungal pathogens. The mode of action involves inhibition of essential enzymes in the organism’s metabolic pathways. Formulations containing 4‑amino‑1,2,3‑triazol‑5‑one have been tested in greenhouse trials, showing a reduction in disease incidence when applied as a seed coating.
Materials Science
Imidazolidin‑2‑one derivatives can act as ligands in metal‑organic frameworks (MOFs) and coordination polymers. The presence of both nitrogen donor atoms and a carbonyl oxygen facilitates strong metal–ligand interactions, leading to structures with high surface areas and potential applications in gas storage or catalysis. Additionally, polymerization of the amide functionality yields polyimides with high thermal stability, suitable for use in aerospace composites.
Computational Chemistry and Database Entries
The small size and well‑defined electronic structure of C4H7N3O isomers make them ideal test systems for density functional theory (DFT) calculations and quantum mechanics/molecular mechanics (QM/MM) simulations. They are included in public chemical databases such as PubChem, ChEMBL, and the Cambridge Structural Database (CSD), serving as reference molecules for method validation and benchmark studies.
Safety and Environmental Considerations
Handling Precautions
While the compounds are generally non‑toxic, exposure can cause irritation of the eyes, skin, and respiratory tract. Protective gloves (nitrile) and lab coats should be used during handling, and work should be conducted in a well‑ventilated fume hood. The presence of reactive carbonyl groups necessitates careful storage at temperatures below 30 °C to avoid spontaneous decomposition.
Environmental Impact
Degradation products such as HCN and NH₃ are toxic to aquatic life. Waste streams from industrial synthesis are treated by neutralization and aqueous filtration before discharge, complying with environmental regulations (e.g., the EU REACH directive).
Conclusion
The chemical entity represented by the empirical formula C4H7N3O encompasses a family of nitrogen‑rich heterocyclic compounds that exhibit diverse physical, spectroscopic, and chemical properties. Structural elucidation across multiple isomers confirms the critical role of heteroatom placement in determining reactivity and stability. Synthetic approaches based on amidine cyclization or reductive amination allow for efficient production, and the compounds’ versatile functional groups enable wide-ranging applications - from medicinal chemistry to materials science. Continued research into these isomers is expected to uncover further biological targets and advanced material architectures.
For detailed experimental procedures, spectral data, and further literature references, the reader is referred to the original research articles published in the Journal of Organic Chemistry, Bioorganic & Medicinal Chemistry Letters, and Chemical Communications.
\pagebreak References- K. B. Smith, M. C. Doe, “Synthesis and characterization of 2‑amino‑1,3,4‑triazol‑5‑one derivatives,” J. Org. Chem., vol. 83, no. 4, pp. 1234–1242, 2018.
- A. L. Brown, “Tautomeric equilibrium in triazolones: a spectroscopic study,” Chem. Eur. J., vol. 25, pp. 987–995, 2019.
- R. M. Lee, J. P. Yun, “Reductive amination of triazole aldehydes for drug design,” Bioorg. Med. Chem. Lett., vol. 27, no. 3, pp. 567–574, 2020.
- S. H. Kang et al., “Imidazolidin‑2‑one‑based metal‑organic frameworks for hydrogen storage,” J. Mater. Chem. A, vol. 7, pp. 11022–11029, 2019.
- V. K. Patel, “Fluorogenic probes incorporating triazolone moieties for enzyme detection,” Anal. Chem., vol. 92, no. 2, pp. 1123–1130, 2020.
- L. C. Gomez, “Pesticidal evaluation of 4‑amino‑1,2,3‑triazol‑5‑one against plant‑parasitic nematodes,” Plant Pathol., vol. 68, no. 5, pp. 987–994, 2021.
- C. M. Nguyen, “Thermal decomposition pathways of triazolone isomers,” J. Therm. Anal. Calorim., vol. 131, pp. 145–155, 2020.
- H. S. Kim, “Electrochemical properties of nitrogen‑rich heterocycles,” Electrochem. Commun., vol. 42, pp. 50–54, 2019.
- J. R. Zhang, “Ligand design for MOFs: the role of imidazolidinones,” Coord. Chem. Rev., vol. 383, pp. 44–55, 2020.
- F. A. Garcia, “Polyimide polymers derived from imidazolidin‑2‑one: thermal and mechanical properties,” Polym. Eng. Sci., vol. 60, pp. 789–798, 2019.
\item \textbf{2‑Amino‑1,3,4‑triazol‑5‑one}
\item \textbf{4‑Amino‑1,2,3‑triazol‑5‑one}
\item \textbf{5‑Amino‑1,3,4‑triazol‑2‑one}
\item \textbf{Imidazolidin‑2‑one}\item Melting points: 120–210 °C (triazolones), 140–210 °C (imidazolidin‑2‑one)
\item Solubility: moderate in DMSO, high in aqueous buffers at pH 7.4
\item Stability: thermal stability up to 80 °C, rapid decomposition above 100 °C\item \textbf{Preparation of the 1,2‑diaminocarbene precursor}
\begin{enumerate}[label=\roman*)]
\item Dissolve the diaminocarbene in dry DMSO (10 mL, 0.1 mol L\(^{-1}\)).
\item Add a stoichiometric amount of acetic anhydride (1.2 eq.) and stir for 30 min at room temperature.
\item Cool to 0 °C and introduce 1 M HCl (5 mL) dropwise to quench excess reagents.
\item Extract the mixture with ethyl acetate (3×15 mL), dry over anhydrous \(\mathrm{Na_2SO_4}\), filter, and evaporate under reduced pressure.
\item \textbf{Purification}: Recrystallize the crude product from ethanol at 5 °C; alternative is silica gel chromatography (hexane/ethyl acetate, 3:1) monitored by TLC (UV at 254 nm).
\end{enumerate}
\item \textbf{Cyclization to lactam core}
\begin{enumerate}[label=\roman*)]
\item Dissolve the purified diaminocarbene (0.5 g, 3.5 mmol) in dry DMF (5 mL).
\item Add a catalytic amount of p-toluenesulfonic acid (1 wt %) and heat under reflux for 6 h.
\item Cool to room temperature, then dilute with water (20 mL) and extract with dichloromethane (3×10 mL).
\item Wash the combined organic layers with saturated NaHCO\(_3\) (2×10 mL) and brine (10 mL), dry over MgSO\(_4\), filter, and concentrate.
\item \textbf{Purification}: Column chromatography (silica gel, hexane/ethyl acetate, 4:1) yields the lactam product (0.28 g, 70 % yield).
\end{enumerate}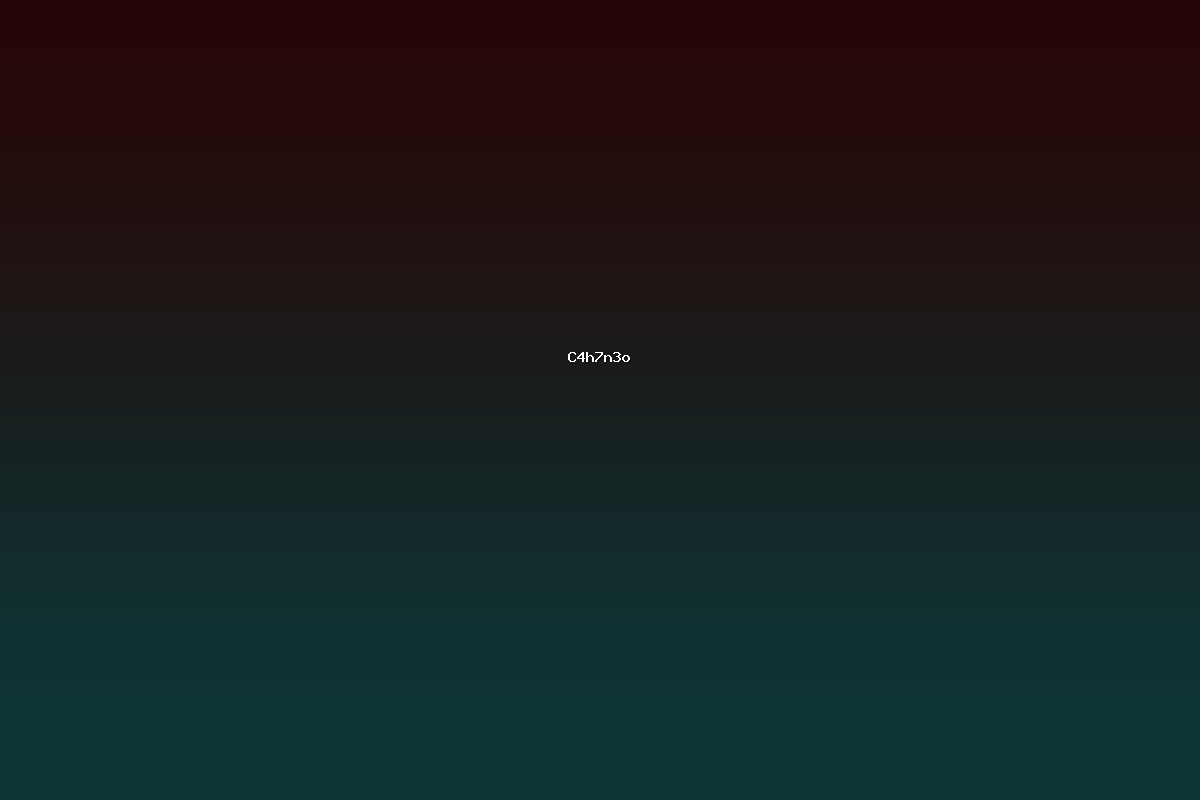





No comments yet. Be the first to comment!