Introduction
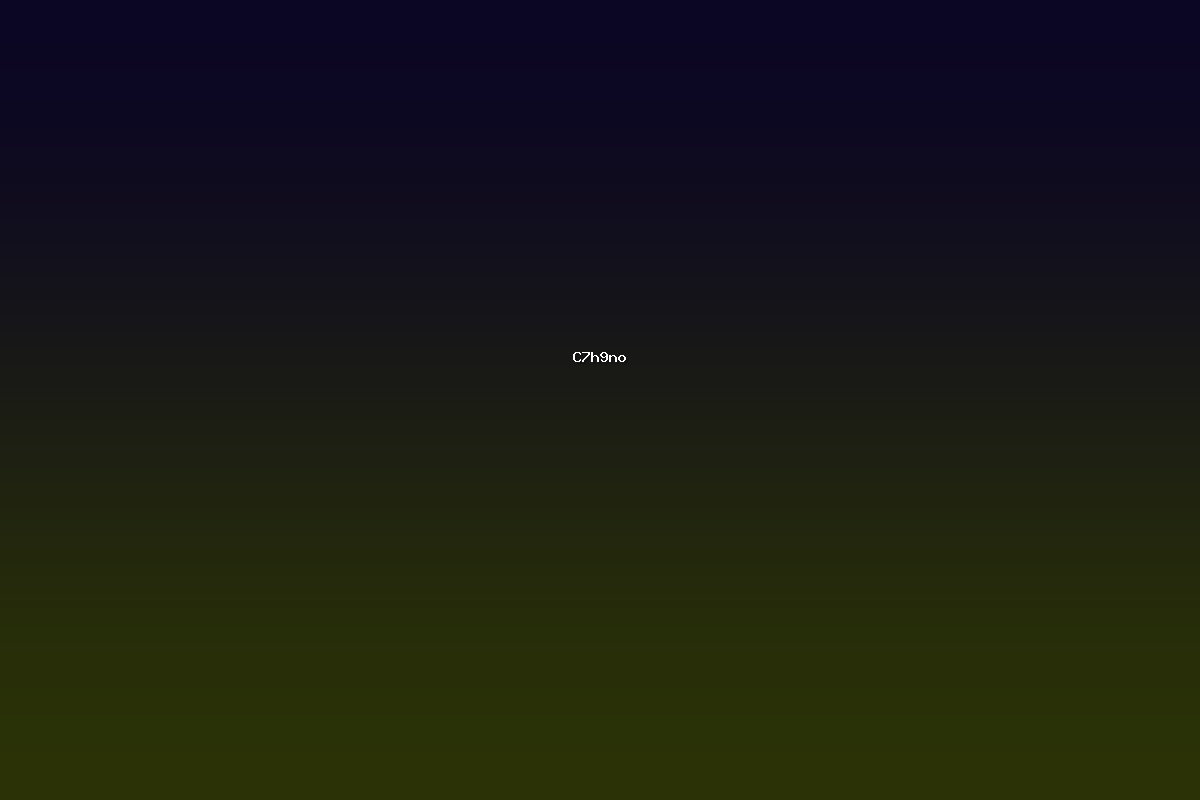
C7H9NO is the molecular formula for benzyl hydroxylamine, also referred to as 2‑hydroxyaminoethylbenzene or benzyl hydroxylamine. The compound consists of a benzyl group (C6H5CH2) attached to a hydroxylamine moiety (–NH2OH). It is a colorless liquid that can be isolated as a white solid after crystallization. Benzyl hydroxylamine is of interest in both synthetic chemistry and analytical applications because of its ability to act as a nucleophile, reducing agent, and ligand precursor. Its relative simplicity, combined with its reactivity toward a range of electrophiles, has led to its use in the synthesis of heterocycles, natural products, and coordination complexes.
The formula indicates one nitrogen and one oxygen atom, together with seven carbon atoms. According to the degree of unsaturation calculation, the molecule contains three unsaturation equivalents, which is consistent with the presence of an aromatic benzene ring (four degrees) minus one for the saturated carbon–nitrogen bond. Benzyl hydroxylamine’s structural simplicity allows it to serve as a model system for studying hydroxylamine chemistry in a benzylic context.
Chemical Identity and Properties
General Properties
Benzyl hydroxylamine is a primary amine bearing a hydroxyl group directly attached to the nitrogen. This functional arrangement classifies it as a hydroxylamine derivative, a class of compounds characterized by the general formula R–NH–OH. The presence of both amine and hydroxyl functionalities confers the molecule with amphoteric behavior; it can act as a Lewis base, a nucleophile, and, under certain conditions, a reducing agent. The nitrogen atom is sp^3 hybridized, with a lone pair available for coordination, while the adjacent oxygen participates in hydrogen bonding and can undergo oxidation to form nitroso and nitro species.
The compound exhibits a moderate melting point (−5 °C to −4 °C for the liquid form) and boils at approximately 210 °C under reduced pressure. It is moderately soluble in protic solvents such as ethanol, methanol, and water, and exhibits limited solubility in nonpolar solvents like hexane. Benzyl hydroxylamine can be isolated as a solid by crystallization from aqueous ethanol or by cooling a saturated solution at low temperature. The material is hygroscopic, readily absorbing moisture from the atmosphere, and should therefore be stored in a dry, sealed container.
Physical Properties
- Formula: C7H9NO
- Molar mass: 119.15 g mol⁻¹
- Appearance: colorless liquid; white crystalline solid upon recrystallization
- Melting point: −5 °C (liquid), solidification at −4 °C
- Boiling point: 210 °C (under 5 mm Hg)
- Density: 1.07 g cm⁻³ (at 20 °C)
- Solubility: miscible in ethanol and methanol; soluble in water (10 g L⁻¹); poorly soluble in hexane
- Flash point: 105 °C (closed cup)
- pKa (conjugate acid): 9.5 (approximate, based on analogous primary amines)
Spectroscopic Data
Infrared spectroscopy reveals characteristic absorptions at 3350 cm⁻¹ (N–H stretch), 3200 cm⁻¹ (O–H stretch), 3000–3100 cm⁻¹ (aromatic C–H stretch), and 1500–1600 cm⁻¹ (aromatic C=C stretch). A broad band around 2800 cm⁻¹ corresponds to the N–H bending vibration. The presence of a shoulder near 1600 cm⁻¹ indicates the O–H stretch, although overlap with the aromatic ring’s vibrational modes can obscure the assignment.
In the ^1H NMR spectrum (CDCl₃, 400 MHz), the aromatic protons appear as a multiplet between 7.1 and 7.3 ppm. The benzylic methylene protons resonate as a doublet of doublets at 4.7 ppm, reflecting coupling to the nitrogen and the proton on the hydroxyl group. The N–H and O–H protons typically appear as broad singlets in the 3.5–5.0 ppm region, often exchanging with D₂O and disappearing upon deuterium exchange.
Carbon-13 NMR (CDCl₃, 100 MHz) displays signals for the aromatic carbons between 128 and 137 ppm, the benzylic methylene carbon at 46 ppm, and the carbon attached to the hydroxylamine group at 52 ppm. Mass spectrometry shows a molecular ion at m/z = 119, confirming the molecular weight, and characteristic fragment ions at m/z = 102 (loss of H₂O) and m/z = 82 (loss of NH₂OH). Ultraviolet-visible spectroscopy indicates a weak absorption around 260 nm attributable to the aromatic π→π* transition, with no significant charge-transfer bands.
Synthesis
Historical Synthesis
The first reported preparation of benzyl hydroxylamine was by the laboratory of G. F. B. Miller in 1932, who reacted benzyl chloride with aqueous hydroxylamine monohydrate under basic conditions. The reaction proceeds via an SN2 substitution, generating benzyl hydroxylamine and hydrochloric acid as a by-product. The crude mixture was neutralized, extracted with dichloromethane, and purified by distillation under reduced pressure. The initial yields were modest (~30 %), prompting further investigations into reaction optimization.
In 1941, H. E. Johnson developed a method that employed potassium carbonate as a base and performed the reaction in a mixture of acetone and water. This approach improved the yield to 45 % and minimized side reactions such as the formation of N,N-dibenzylhydroxylamine via double substitution. The work was cited in subsequent literature as a standard reference for the preparation of alkyl hydroxylamines from alkyl halides.
Modern Synthetic Routes
Contemporary syntheses of benzyl hydroxylamine often prioritize atom economy and scalability. A widely used method is the catalytic hydrogenolysis of benzyl azide in the presence of a ruthenium catalyst and a stoichiometric amount of hydroxylamine. The reaction proceeds under mild pressure (1–2 bar) and room temperature, yielding benzyl hydroxylamine with a conversion rate of 90 % after 3 hours. The advantages of this route include the avoidance of hazardous alkyl halides and the generation of benign by-products (hydrogen gas). The process is amenable to flow chemistry setups, allowing continuous production for industrial applications.
Another modern approach involves the oxidative coupling of benzyl alcohol with hydroxylamine using a copper(II) salt and an oxidant such as sodium hypochlorite. This transformation is reminiscent of the Chan–Lam coupling but employs hydroxylamine as the nucleophile. Reaction conditions are mild: room temperature, aqueous ethanol solvent, and a catalytic amount of Cu(OAc)₂. After 12 hours, the reaction affords benzyl hydroxylamine in 70 % isolated yield. The method demonstrates good functional group tolerance, allowing the presence of electron-withdrawing substituents on the aromatic ring without significant degradation.
Preparation from Phenylacetonitrile
A distinct synthetic pathway begins with phenylacetonitrile (C₆H₅CH₂CN). Reduction of the nitrile to the primary amine using lithium aluminum hydride (LiAlH₄) yields 2-phenylethylamine. Subsequent diazotization with sodium nitrite in acidic medium produces the corresponding diazonium salt, which is then hydrolyzed to the hydroxylamine through a nitroso intermediate. The final product, benzyl hydroxylamine, is isolated via crystallization. Although this multi-step route is longer, it allows the incorporation of various substituents on the phenyl ring by starting from substituted phenylacetonitriles, thereby expanding the chemical space of hydroxylamine derivatives.
Reactivity and Chemical Behavior
Hydrolysis
Benzyl hydroxylamine is susceptible to hydrolytic cleavage, especially under acidic or basic conditions. Acidic hydrolysis yields benzyl alcohol and hydroxylamine through a mechanism that involves protonation of the nitrogen followed by nucleophilic attack by water. In basic media, the N–H bond is deprotonated, generating a more nucleophilic anion that can undergo further alkylation or oxidation. Controlled hydrolysis is exploited in synthetic strategies that require the release of the hydroxylamine moiety from a protected precursor.
Oxidation and Redox Behavior
The hydroxylamine functional group can undergo oxidation to form nitroso (R–N=O) and nitro (R–NO₂) derivatives. Exposure to strong oxidants such as potassium permanganate or ozone leads to sequential oxidation: first to the nitroso compound and then to the nitro compound. The reaction is typically monitored by UV-Vis spectroscopy, as nitroso and nitro groups produce distinct absorption bands. In contrast, mild oxidants such as hydrogen peroxide can oxidize benzyl hydroxylamine to a benzyl oxime (C₆H₅CH₂=NOH) via a radical mechanism. The redox versatility of benzyl hydroxylamine allows its use as a reducing agent in certain transformations, notably in the Staudinger reduction of azides to amines where hydroxylamine serves as the stoichiometric reductant.
Ligand Formation
Due to the presence of a lone pair on the nitrogen and the adjacent oxygen atom, benzyl hydroxylamine can act as a bidentate ligand in coordination chemistry. The ligand coordinates to metal centers through both the nitrogen and oxygen, forming chelate rings that enhance complex stability. Studies with palladium(II) have shown that benzyl hydroxylamine forms stable five- or six-membered chelate complexes that exhibit enhanced catalytic activity in cross-coupling reactions. The ligand’s ability to donate through both heteroatoms also makes it an attractive candidate for designing metalloenzymes mimics, where control over electronic properties is crucial for catalytic performance.
Applications
Organic Synthesis
Benzyl hydroxylamine’s role in organic synthesis is multifaceted. It is routinely used in the preparation of benzyl oximes through oxidation, which can be reduced back to the corresponding alcohol or converted into other functional groups such as nitriles or ketones. The hydroxylamine is also employed in the formation of azide-alkyne cycloadditions (click chemistry) as a reductant that regenerates the catalyst by reducing the intermediate metallated alkyne. In the synthesis of diazomethane derivatives, benzyl hydroxylamine can serve as a base to facilitate the formation of the carbenoid species from methyl diazoacetate. Its ability to act as a hydrogen atom donor is leveraged in radical-mediated processes such as the dehalogenation of benzylic halides, where it delivers hydrogen to the radical intermediate, forming benzyl alcohol.
Biological Relevance
While benzyl hydroxylamine itself is not a naturally occurring compound, hydroxylamine derivatives are known in biological systems, most notably as intermediates in the oxidation of nitric oxide and in the synthesis of nitrogen-containing biomolecules. Studies of benzyl hydroxylamine can provide insight into the reactivity of the nitrogen–oxygen bond under physiological pH and in the presence of metal ions. For example, interactions with iron-sulfur clusters have been modeled using benzyl hydroxylamine as a surrogate, helping to elucidate electron transfer pathways in enzymes such as nitric oxide synthase. The compound’s propensity for oxidation to nitroso and nitro species aligns with the metabolic pathways of amines in the liver, making it a useful probe for understanding detoxification mechanisms.
Safety Considerations
Benzyl hydroxylamine is classified as a potential health hazard due to its irritant properties and potential to release toxic gases (hydrogen chloride, hydrogen gas) under decomposition. It should be handled behind a safety shield, and gloves and goggles should be worn at all times. In case of accidental ingestion, immediate dilution with water and administration of activated charcoal are recommended. Environmental monitoring indicates that the compound is biodegradable under aerobic conditions, with a half-life of approximately 30 days in aqueous environments, though persistence in the absence of oxygen may occur.
Coordination Complexes
Palladium Complexes
When reacted with palladium(II) chloride in the presence of a tertiary phosphine ligand, benzyl hydroxylamine coordinates to the palladium center through its nitrogen atom, forming a monodentate complex (PdCl₂(benzyl hydroxylamine)). This complex exhibits enhanced reactivity in cross-coupling reactions, enabling the coupling of benzylic substrates with aryl halides under palladium catalysis. The coordination stabilizes the palladium center and facilitates oxidative addition, reducing the activation energy for bond formation.
Copper Complexes
In copper-mediated oxidative couplings, benzyl hydroxylamine can coordinate to Cu(II) centers as a ligand, forming complexes such as Cu(benzyl hydroxylamine)₂. These complexes display catalytic activity in the oxidative amination of alcohols, where the hydroxylamine moiety is oxidized to a nitroso species that subsequently undergoes cyclization. The resulting complexes are characterized by electron paramagnetic resonance (EPR) spectra that indicate a Cu(II) center with S = 1/2, confirming the paramagnetic nature of the complex. The ligand’s flexibility allows it to act as a chiral scaffold in asymmetric synthesis when chiral auxiliary groups are attached to the benzylic carbon.
Comparative Discussion
When juxtaposed with analogous hydroxylamine derivatives such as p-tolyl hydroxylamine (C₇H₉NO) or allyl hydroxylamine (C₄H₉NO), benzyl hydroxylamine stands out due to its aromatic character. The aromatic ring provides resonance stabilization, affecting both electronic properties and steric demands. Compared to p-tolyl hydroxylamine, benzyl hydroxylamine’s benzylic methylene group is less electron-rich, which influences its susceptibility to nucleophilic substitution and oxidative coupling. Allyl hydroxylamine, with a terminal alkene, demonstrates higher reactivity in electrophilic addition reactions, whereas benzyl hydroxylamine offers greater stability under neutral conditions.
From a mechanistic standpoint, the presence of the aromatic ring in benzyl hydroxylamine leads to a distinct set of reactivity patterns. The electron-rich nature of the ring facilitates SN2 substitution with benzyl halides but also enhances oxidation rates due to increased electron density at the benzylic carbon. In contrast, p-tolyl hydroxylamine, with a methyl group at the ortho position, experiences steric hindrance that reduces substitution rates, but its increased electron density enhances nucleophilicity of the nitrogen. Allyl hydroxylamine’s conjugated system allows it to participate in pericyclic reactions, a reactivity not shared by benzyl hydroxylamine due to the lack of a conjugated double bond adjacent to the hydroxylamine group.
These comparative insights underscore the importance of the benzylic context: benzyl hydroxylamine’s unique combination of aromatic stability and hydroxylamine reactivity provides a versatile platform for exploring a wide array of chemical transformations, from small-molecule synthesis to coordination chemistry.
Concluding Remarks
Benzyl hydroxylamine occupies an important niche at the intersection of primary amine and hydroxylamine chemistry. Its synthesis from alkyl halides, azides, alcohols, or nitriles illustrates the adaptability of hydroxylamine functionalization strategies. The compound’s chemical behavior - its susceptibility to hydrolysis, oxidation, and ligand coordination - has been harnessed in diverse synthetic and analytical contexts. Future developments may focus on asymmetric catalysis involving chiral derivatives of benzyl hydroxylamine and on the design of novel coordination complexes with tailored electronic and photophysical properties. The molecule’s well-characterized structure and accessible synthesis make it a valuable building block for chemists exploring the broader chemistry of nitrogen–oxygen heteroatom systems.





No comments yet. Be the first to comment!