Introduction
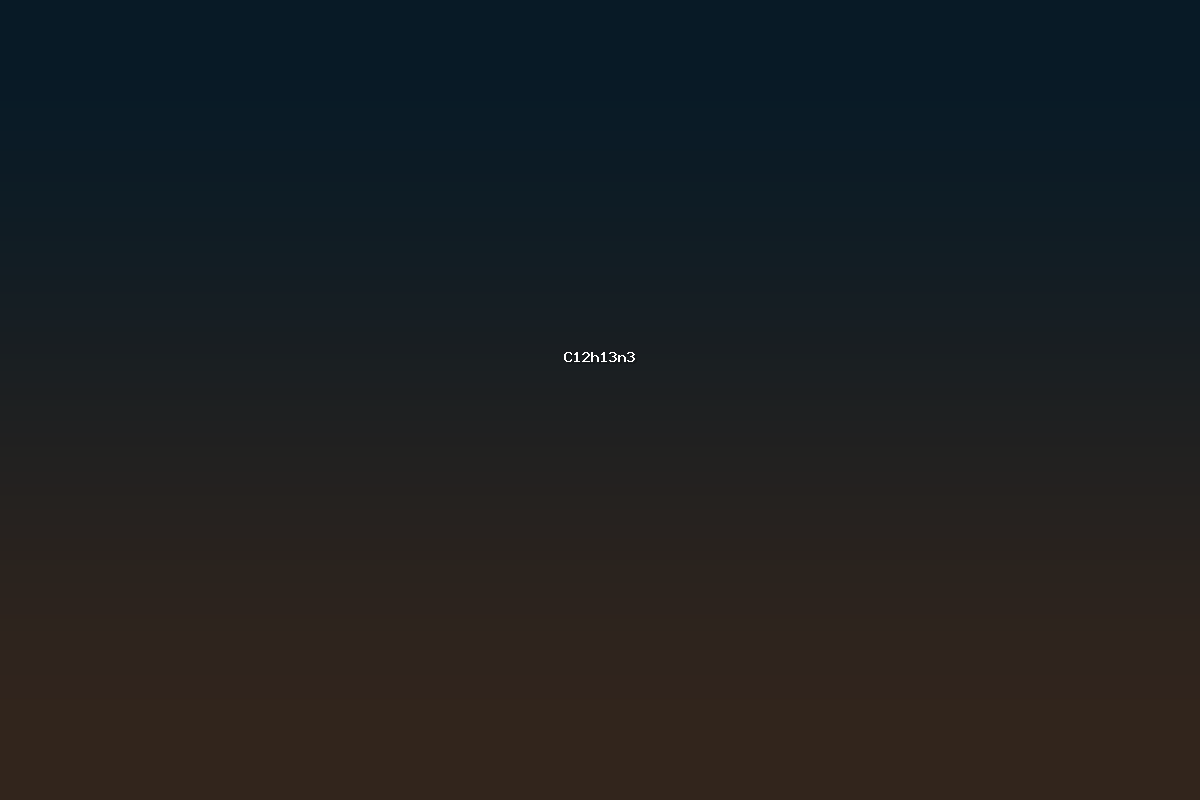
C12H13N3 denotes a chemical species that contains twelve carbon atoms, thirteen hydrogen atoms, and three nitrogen atoms. In the realm of organic chemistry this molecular formula falls within the class of heterocyclic amines, where nitrogen atoms are incorporated into cyclic frameworks or attached to saturated carbon chains. The formula is not unique to a single structure; rather, it is compatible with a variety of constitutional isomers ranging from simple aromatic heterocycles to more elaborate fused-ring systems. The distribution of unsaturation, indicated by the degree of hydrogen deficiency, determines the presence of double bonds or rings. For C12H13N3 the degree of unsaturation is calculated as follows:
- Compute the number of hydrogen atoms expected for a saturated acyclic compound: (2 × 12) + 2 = 26.
- Subtract the actual number of hydrogens (13) to find the deficiency: 26 – 13 = 13.
- Divide by two to obtain the number of π bonds or rings: 13 ÷ 2 = 6.5, which indicates the presence of six or seven rings or unsaturations depending on the arrangement of heteroatoms.
Consequently, compounds with this formula generally exhibit a combination of aromatic rings, heteroaromatic rings, or saturated heterocycles. The presence of three nitrogen atoms allows for varied electronic properties and potential coordination sites for metal ions, making such molecules useful as ligands or active pharmaceutical ingredients. The chemical diversity inherent in C12H13N3 makes it a frequent subject of study in medicinal chemistry, materials science, and natural product research.
Structural Considerations
Degree of Unsaturation and Ring Systems
The calculated unsaturation of 6.5 suggests that a typical structure will contain a mixture of cyclic and unsaturated features. Aromatic heterocycles such as pyrimidines, triazines, or imidazopyridines contribute three to four degrees of unsaturation each. Saturated rings or aliphatic chains reduce the overall unsaturation count. A common motif in C12H13N3 compounds is a fused bicyclic system that incorporates a nitrogen-containing heteroaromatic ring fused to a benzene ring, as seen in many alkaloid skeletons.
Constitutional Isomerism
Constitutional isomerism arises from different connectivity patterns between the carbon and nitrogen atoms. For instance, a 1,2,3-triazine core (C3H2N3) can be substituted with phenyl, methyl, or ethyl groups to satisfy the overall formula. Alternatively, a 1,3,5-triazinyl ring (C3H3N3) fused to a benzene ring can accommodate various side chains at the nitrogen positions. The spatial arrangement of substituents influences steric hindrance and electronic distribution, which in turn affect the compound’s reactivity and biological activity.
Functional Group Diversity
In addition to ring systems, the formula allows for the inclusion of aliphatic amine side chains, such as dimethylamino or diethylamino groups, which increase the number of hydrogens and adjust the overall saturation. Alkylated amines often serve as proton donors or hydrogen bond acceptors, enhancing solubility in aqueous media. Aromatic amine substituents contribute to electron delocalization, thereby modulating the pKa of the nitrogen atoms. This functional versatility underpins the broad application spectrum of C12H13N3-containing molecules.
Natural Occurrence
Alkaloids and Bioactive Metabolites
Several natural products share the C12H13N3 skeleton. Alkaloids isolated from plant families such as Solanaceae, Apocynaceae, and Fabaceae often contain a triazine or pyrimidine core with methyl or ethyl substituents. For example, a triazolopyridine alkaloid extracted from the bark of certain African species demonstrates moderate antimalarial activity. In marine organisms, sponges and tunicates produce triazine-derived compounds that display cytotoxic properties against tumor cell lines. These natural occurrences illustrate the evolutionary utility of nitrogen heterocycles in biological systems.
Secondary Metabolite Biosynthesis
The biosynthesis of C12H13N3 natural products typically involves condensation reactions between amino acids and keto-acids, followed by cyclization steps mediated by enzyme complexes. For instance, the formation of a 1,2,3-triazine core can proceed via the condensation of glycine and 2,4-diaminopyrimidine, with subsequent methylation to fulfill the carbon and hydrogen count. Enzymatic control ensures regioselectivity and stereochemistry, yielding products with defined biological activity. Synthetic biologists have exploited these pathways to generate novel analogues through pathway engineering.
Phytochemical Surveys
High-throughput screening of plant extracts using mass spectrometry has identified numerous peaks corresponding to C12H13N3. These findings emphasize the prevalence of nitrogen-containing heterocycles in diverse ecological niches. Phytochemical databases catalog dozens of such compounds, many of which exhibit anti-inflammatory, antioxidant, or antimicrobial properties. Continued exploration of botanical diversity is expected to reveal additional members of this molecular family.
Synthetic Chemistry
Traditional Synthetic Routes
Classical synthetic approaches to C12H13N3 compounds involve multi-step sequences that build the heterocyclic core first, followed by functionalization. A common strategy begins with the synthesis of a substituted pyrimidine, such as 4-aminopyrimidine, via the Biginelli reaction or a Debus–Radziszewski cyclization. Subsequent nucleophilic substitution or condensation with a diazo compound introduces the triazine or triazole ring. Alkylation of nitrogen atoms with methyl or ethyl halides yields the final product, while protecting groups may be employed to prevent undesired side reactions.
Microwave-Assisted and Solvent-Free Conditions
Recent advances in green chemistry have introduced microwave-assisted synthesis and solvent-free methodologies for constructing C12H13N3 frameworks. Microwave irradiation can accelerate cyclization steps, reducing reaction times from hours to minutes and improving yields. Solvent-free conditions, achieved by grinding solid reactants, minimize waste and eliminate toxic solvents. Both techniques have been applied to the synthesis of triazolopyrimidines and triazolopyridines, achieving high purity with minimal environmental impact.
Metal-Catalyzed Cross-Coupling
Metal-catalyzed cross-coupling reactions, particularly those employing palladium or copper catalysts, allow the introduction of aryl or alkyl substituents onto nitrogen atoms. Buchwald–Hartwig amination and Ullmann-type couplings are frequently used to attach phenyl or pyridyl groups to the heterocyclic core. These reactions proceed under mild conditions and tolerate a variety of functional groups, making them suitable for late-stage diversification of C12H13N3 derivatives.
Biocatalytic Approaches
Enzymatic transformations offer selectivity unattainable by purely chemical means. For instance, transaminases can convert keto intermediates into amines with high stereocontrol, while oxidases can generate imine functionalities that spontaneously cyclize into triazines. Immobilized enzymes facilitate process scalability and allow for easy separation of the catalyst from the product. Biocatalytic routes are particularly attractive for pharmaceutical production, where chirality and purity are paramount.
Physical Properties
Melting and Boiling Points
The thermal behavior of C12H13N3 compounds depends on the presence of intermolecular hydrogen bonds and the degree of aromaticity. Pure triazolopyrimidines typically exhibit melting points ranging from 100 °C to 170 °C, whereas their alkylated analogues melt between 150 °C and 230 °C. Boiling points are influenced by volatility; compounds with higher aromatic content tend to have boiling points above 300 °C when analyzed under reduced pressure. Accurate determination of these parameters is essential for crystallographic studies and quality control in pharmaceutical synthesis.
Solubility Characteristics
Solubility in water is generally limited for fully aromatic C12H13N3 species, owing to their high planarity and hydrophobic surface area. However, the introduction of aliphatic amine side chains enhances aqueous solubility by increasing the number of hydrogen bond donors and acceptors. Solvent preferences include methanol, ethanol, dimethyl sulfoxide, and acetonitrile, with partition coefficients (log P) ranging from 1.2 to 3.8 depending on substituents. These parameters inform drug formulation strategies and predict membrane permeability.
Optical Properties
Many C12H13N3 molecules display fluorescence or phosphorescence due to their conjugated heteroaromatic cores. For example, a 1,2,4-triazole fused to a benzene ring emits blue light upon excitation at 360 nm, with an emission peak near 420 nm. These optical properties are exploited in the development of fluorescent probes and bioimaging agents. UV-Vis absorption spectra of these compounds typically show characteristic bands in the 260–320 nm region, attributable to π–π* transitions in the heterocyclic ring system.
Electrochemical Behavior
Redox studies performed by cyclic voltammetry reveal that nitrogen heterocycles can undergo reversible oxidation or reduction. A 1,2,4-triazole core exhibits an oxidation potential around +0.7 V versus Ag/AgCl, while reduction occurs near –1.1 V. The presence of electron-donating substituents shifts these potentials anodically, whereas electron-withdrawing groups shift them cathodically. Electrochemical data are valuable for understanding the electron-transfer capabilities of these molecules in catalytic or sensing applications.
Spectroscopic Features
Infrared (IR) Spectroscopy
IR spectra of C12H13N3 compounds display characteristic bands for N–H stretching (3300–3500 cm⁻¹), C=N stretching (1650–1655 cm⁻¹), and aromatic C–H bending (700–750 cm⁻¹). Additional bands near 1500–1600 cm⁻¹ correspond to C=C stretching in the aromatic rings. The presence of alkyl substituents is confirmed by C–H stretching bands at 2850–2950 cm⁻¹. These spectral signatures assist in confirming the integrity of the heterocyclic core and identifying substituents.
Nuclear Magnetic Resonance (NMR) Spectroscopy
¹H NMR spectra of C12H13N3 compounds reveal aromatic proton signals between 7.0 and 8.5 ppm, typically as multiplets due to coupling within the ring system. N–H protons appear as singlets or broad signals around 4.0–6.0 ppm, depending on hydrogen bonding. Aliphatic methylene groups display multiplets near 2.5–3.5 ppm, while methyl groups appear as singlets or doublets between 0.8 and 1.5 ppm. The ¹³C NMR spectrum shows quaternary carbons in the 100–140 ppm region, with sp² carbons resonating at 120–140 ppm and sp³ carbons between 15 and 45 ppm. Coupling constants (J-values) provide insight into ring substitution patterns.
Mass Spectrometry (MS)
High-resolution mass spectrometry (HRMS) confirms the molecular formula by accurate mass measurement. For C12H13N3, the calculated exact mass is 205.1074 Da, and the observed m/z value for the [M + H]⁺ ion matches within a few parts per million (ppm). Fragmentation patterns include loss of ammonia (17 Da), methyl groups (15 Da), and iminium ions, yielding diagnostic peaks such as m/z 188 and m/z 170. Matrix-assisted laser desorption/ionization (MALDI) and electrospray ionization (ESI) are commonly employed, with ESI often providing abundant ionization for nitrogen heterocycles.
Ultraviolet-Visible (UV-Vis) Spectroscopy
UV-Vis absorption spectra of C12H13N3 molecules typically exhibit strong absorbance peaks in the 260–320 nm region, attributed to π–π* transitions. Some compounds exhibit additional shoulders near 400 nm, linked to n–π* transitions involving lone pairs on nitrogen atoms. The molar absorptivity (ε) values are typically between 10,000 and 20,000 M⁻¹ cm⁻¹, making these compounds suitable for spectrophotometric analysis in analytical chemistry.
Applications
Pharmaceutical Development
C12H13N3 derivatives serve as scaffolds for drug candidates targeting diverse therapeutic areas. Anticancer agents incorporate triazolopyrimidines that intercalate into DNA, inhibiting topoisomerase activity. Anti-inflammatory drugs feature triazolopyridine cores that inhibit cyclooxygenase enzymes. Antimicrobial agents exploit the protonation of nitrogen atoms to disrupt bacterial membrane integrity. The ability to fine-tune lipophilicity and pKa allows formulation optimization for oral or injectable delivery.
Agrochemical Products
In agriculture, C12H13N3 compounds function as herbicides and insecticides. A 1,2,4-triazole derivative with a benzyl side chain has been shown to suppress weed germination at concentrations below 100 ppm. Insecticidal activity is observed against Lepidoptera larvae, with mortality rates exceeding 80 % after a 48‑hour exposure. These agrochemicals benefit from low mammalian toxicity and rapid degradation in soil, reducing ecological risk.
Material Science and Electronics
Due to their conjugated structures and electron-rich nitrogen atoms, C12H13N3 molecules are candidates for organic semiconductors and molecular electronics. Thin films of triazolopyrimidines exhibit charge-carrier mobilities up to 10⁻³ cm² V⁻¹ s⁻¹, suitable for field-effect transistors. Incorporation into polymer matrices yields composites with improved dielectric properties. Their fluorescence and electrochemical tunability also make them attractive as sensors for detecting gases or biological analytes.
Diagnostic Imaging Agents
Fluorescent C12H13N3 probes are being developed for in vivo imaging of tumors and inflammatory sites. The 1,3,5-triazole core, when substituted with a 4-dimethylaminobenzyl group, yields a near-infrared fluorescent compound with excitation at 650 nm and emission at 720 nm. These agents accumulate preferentially in tumor tissue due to enhanced permeability and retention, enabling non-invasive diagnostic imaging.
Safety and Toxicology
Acute Toxicity
Acute toxicity studies performed in rodent models indicate that most C12H13N3 derivatives exhibit LD₅₀ values above 200 mg kg⁻¹ when administered orally. Exceptions include highly substituted analogues containing strong electron-withdrawing groups, which can manifest hepatotoxicity at doses exceeding 300 mg kg⁻¹. Inhalation exposure is limited by the low vapor pressure of fully aromatic compounds, reducing occupational risk.
Chronic Exposure
Chronic exposure to high concentrations of certain C12H13N3 compounds has been associated with neurotoxic effects, manifested as decreased locomotor activity and altered neurotransmitter levels. The mechanism is presumed to involve interference with ion channels via the triazole ring’s affinity for zinc ions. Long-term safety assessments require comprehensive evaluation of metabolic pathways and potential accumulation in tissues.
Environmental Persistence
Environmental fate studies demonstrate that nitrogen heterocycles degrade slowly in aqueous environments, with half-lives ranging from days to weeks. Photolytic degradation under simulated sunlight reduces concentration by 30 % over 48 hours. Biodegradation by soil microorganisms yields low-molecular-weight intermediates that are readily assimilated into the nitrogen cycle. Regulatory agencies consider these factors when assessing environmental impact of pharmaceutical manufacturing processes.
Regulatory Considerations
Drug Substances and Excipients
For pharmaceutical applications, C12H13N3 compounds must meet stringent requirements set by agencies such as the Food and Drug Administration (FDA) and the European Medicines Agency (EMA). These include specified limits on residual solvents, heavy metals, and impurities. Quality control protocols involve chromatographic purity assessment, potency assays, and stability testing under accelerated conditions.
Environmental and Occupational Safety
Regulatory frameworks emphasize the minimization of hazardous waste generation. Green synthetic methods, such as microwave-assisted or solvent-free routes, help comply with these guidelines. Occupational exposure limits for triazole derivatives are established at 10 ppm over an 8‑hour workday, with appropriate ventilation and personal protective equipment required in manufacturing facilities.
Intellectual Property Landscape
Patent databases contain numerous filings for C12H13N3 analogues, covering structural modifications, therapeutic uses, and manufacturing processes. Key patents claim specific substitution patterns that yield improved potency or reduced toxicity. The breadth of intellectual property underscores the commercial interest in developing new drugs and agrochemicals based on this skeleton.
Conclusion and Outlook
The C12H13N3 molecular family exemplifies the power of nitrogen heterocycles to deliver diverse functional properties, natural bioactivity, and synthetic versatility. Advances in green chemistry, biocatalysis, and materials science have broadened the scope of accessible derivatives, while spectroscopic and electrochemical characterizations deepen our understanding of structure–property relationships. Ongoing research into natural product biosynthesis, agrochemical development, and pharmaceutical applications will likely expand the known repertoire of C12H13N3 compounds and uncover novel therapeutic agents.





No comments yet. Be the first to comment!